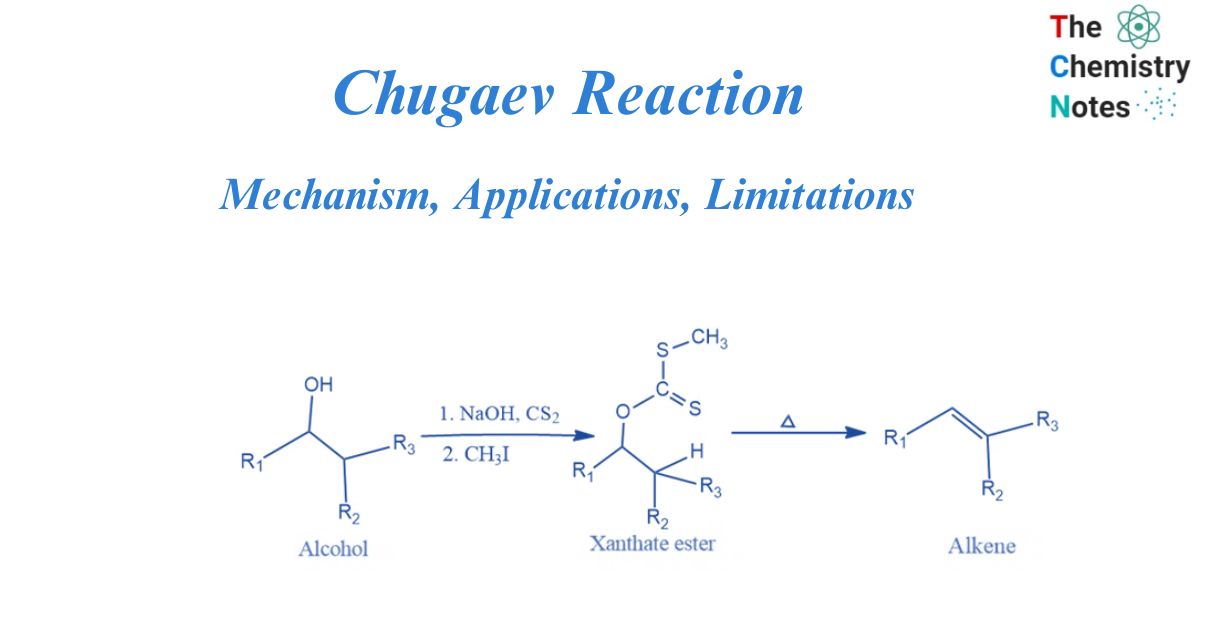
Chugaev reaction is a chemical process that eliminates water from an alcohol, resulting in the formation of an alkene. Xanthate is the intermediate. It is named after Russian chemist Lev Aleksandrovich Chugayev, who described the reaction sequence for the first time in 1899.
In the first stage, an alkoxide and carbon disulfide (CS2) are combined to generate a xanthate salt. Iodomethane is used to convert the alkoxide to methyl xanthate. Alkenes are generated by intramolecular elimination at around 200 °C. A hydrogen atom is transferred from a carbon atom to xanthate oxygen in syn elimination in the transition state of the 6-membered ring.
What is Chugaev’s reaction?
The Chugaev reaction mechanism is a concerted procedure. In this process, sodium xanthate is formed by the reaction of alcohol with carbon disulfide in the presence of sodium hydroxide. Sodium xanthate is then alkylated with methyl iodide to form xanthate ester. When pyrolyzed, this xanthate yields olefin or alkene. Thus, the Chugaev reaction converts alcohol into olefin via the equivalent xanthate esters. This is also known as the Chugaev elimination or the Chugaev xanthate reaction.
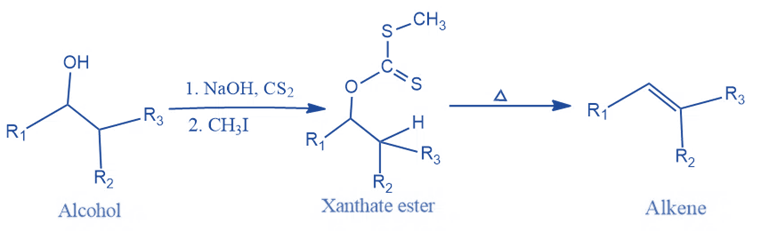
Mechanism of Chugaev Reaction
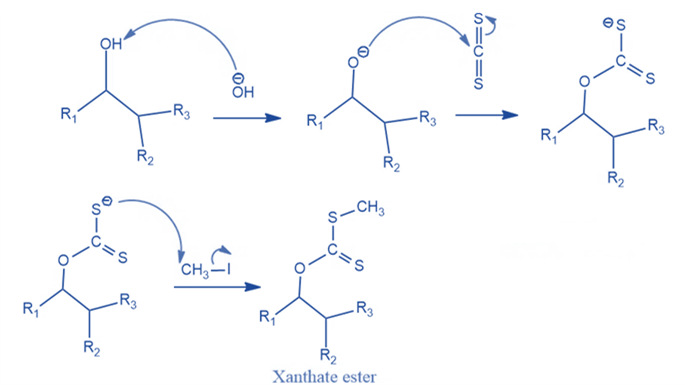
- Formation of xanthate ester
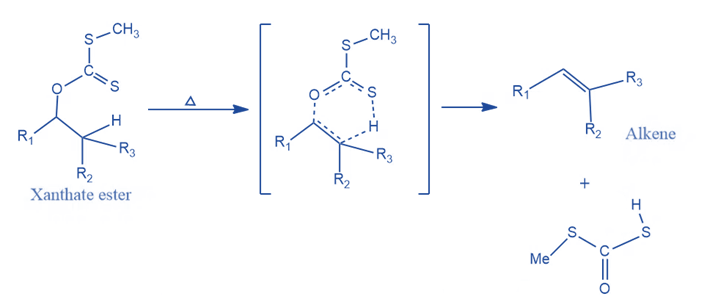
2. Pyrolysis process
Application of Chugaev Reaction
- This reaction is extremely effective for converting secondary and tertiary alcohols into alkenes.
- This reaction can be used to make a variety of olefins such as bornylene, menthene, and others.
Limitations of Chugaev Reaction
- A mixture of isomeric olefins is formed if the Beta-carbon includes more than one hydrogen or if elimination is possible in more than one place.
- It is difficult to break down by heating because the xanthate ester of primary alcohol has excellent thermal stability.
References
- https://chemistry-reaction.com/chugaev-elimination/.
- Wang, Z., Comprehensive Organic Name Reactions and Reagents, John Wiley & Sons, Inc.,2010
- Laue T.,Plagens A.,Named Organic Reactions, Second edition, John Wiley & Sons, Ltd, 2005.
- https://academic-accelerator.com/encyclopedia/chugaev-elimination
