Neodymium is a chemical element with an atomic number of 60 and is represented by the symbol ‘Nd’ in the periodic table. It is hard and silvery in appearance and classified as rare earth metal and belongs to the f-block of the lanthanide group of the periodic table. Neodymium exhibits rapid reactivity upon oxidation, resulting in the formation of compounds with distinct colors, namely pink, purple/blue, and yellow, corresponding to its +2, +3, and +4 oxidation states, respectively.
The Earth’s crust exhibits a neodymium abundance of approximately 38 milligrams per kilogram, positioning it as the 27th most prevalent element. Lanthanum and this element share a comparable level of abundance. Neodymium, in its unbound state, has a limited occurrence in the environment. On the contrary, it is frequently encountered within ores, such as monazite and bastnäsite, which encompass a collective group of minerals instead of individual entities. These ores possess minute quantities of various rare-earth metals, including neodymium.
Interesting Science Videos
History of Neodymium
- The discovery of neodymium took place in 1885 in Vienna, Austria, under the supervision of the renowned Austrian scientist Carl Auer von Welsbach. The substance known as “didymium,” which was mistakenly classified as a new element by Swedish chemist Carl Mosander in 1841, was subsequently found to be of a different nature.
- In the year 1879, the French chemist Lecoq de Boisbaudran successfully identified and isolated the element samarium from the compound known as ‘didymium’ within the city of Paris.
- Following the discovery of samarium, it was observed that the absorption spectrum of ‘didymium’ exhibited variations depending on the specific mineral from which it was derived.
- In the year 1885, Carl Welsbach, the individual credited with the initial discovery of ‘didymium’ fourteen years prior, came to the realization that it was, in fact, a composite of two distinct and previously unknown chemical elements.
Occurrence of Neodymium
- The neodymium abundance in the Earth’s crust is estimated to be about 38 milligrams per kilogram, making it the 27th most abundant element. Lanthanum and neodymium exhibit a similar degree of prevalence.
- The presence of neodymium in its free form is relatively scarce within the natural environment. In contrast, it is commonly found within mineral deposits, such as monazite and bastnäsite.
- The commercial-scale extraction of this substance is accomplished through the application of ion exchange and solvent extraction methodologies, predominantly sourced from monazite and bastnaesite.
- The synthesis of neodymium metal can be accomplished by employing metallic calcium to reduce anhydrous halides.
- Neodymium exhibits a diverse array of isotopes, totaling 30 in number, each possessing well-documented half-lives. These isotopes span a range of mass numbers, specifically from 127Nd to 156Nd. The presence of seven isotopes characterizes naturally-occurring neodymium.
Isotopes of Neodymium
Neodymium has seven naturally occurring isotopes: 142Nd (27.153 %), 143Nd (12.173 %), 144Nd (23.798 %), 145Nd (8.293 %), 146Nd (17.189 %), 148Nd (5.756 %), and 150Nd (5.638 %).
Naturally Occurring Isotopes of Neodymium
| Isotopes | Natural Abundance (atom %) |
|---|---|
| 142Nd | 27.153 |
| 143Nd | 12.173 |
| 144Nd | 23.798 |
| 145Nd | 8.293 |
| 146Nd | 17.189 |
| 148Nd | 5.756 |
| 150Nd | 5.638 |
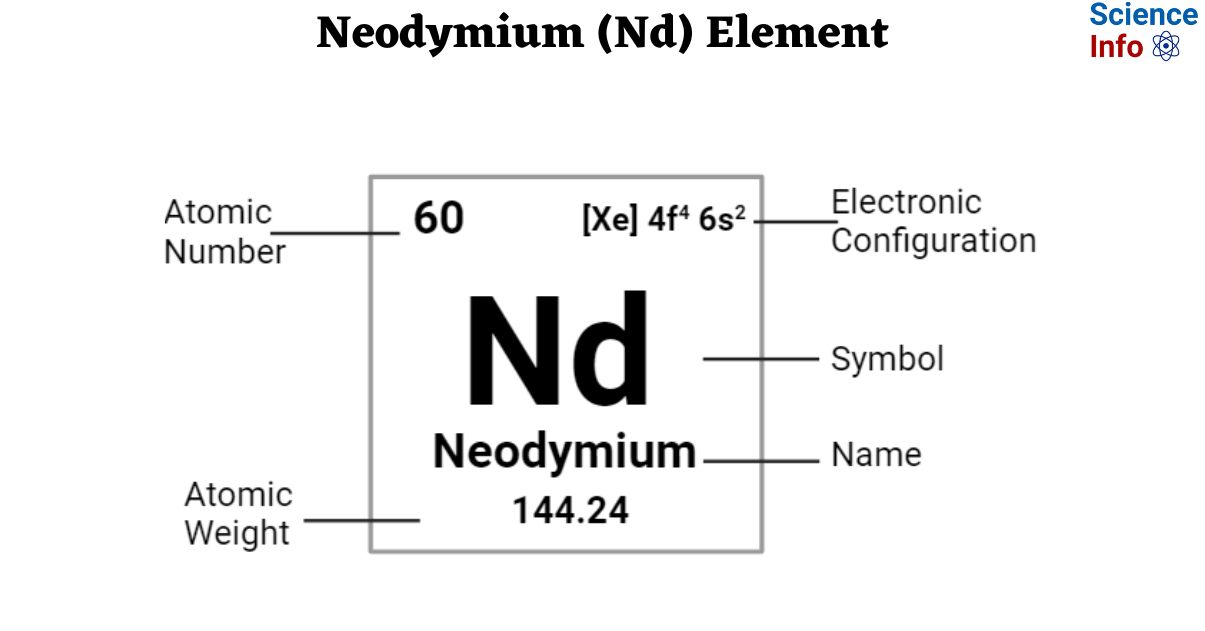
Elemental Properties of Neodymium
| Electronic Configuration | [Xe] 4f4 6s2 |
| Atomic Number | 60 |
| Atomic Weight | 144.24 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | Lanthanides, 6, f-block |
| Density | 7.01 g/cm3 at 20 °C |
| Appearance | silvery-white |
| Van der Waals radius | 0.181 nm |
| Electron shells | 2, 8, 18, 22, 8, 2 |
| Electrons | 60 |
| Protons | 60 |
| Neutrons in the most abundant isotope | 82 |
Physical Properties of Neodymium
- Neodymium has an atomic number of 60 and is a silvery-white rare earth metal. It has a melting point of 1024°C (1875°F) and a boiling point of 3030°C (5,490°F).
- Nd has a solid phase density of 7.01 g/cm3 and a liquid or molten phase density of 6.89 g/cm3.
- It is malleable which means it can be easily beaten into thin sheets without any cleavage.
- It is ductile which means it is possible to draw thin wires from it without breaking.
- At ambient temperature, neodymium exhibits paramagnetic behavior. The material exhibits ferromagnetic properties upon reaching a temperature of −253.2°C.
- Neodymium typically occurs as a trivalent cation, denoted as Nd3+, within its chemical compounds. The majority of its salts exhibit a pale purple hue.
- Neodymium is present in a double hexagonal crystalline structure at temperatures below 863°C. At temperatures exceeding 863°C, neodymium is found to exhibit a body-centered-cubic crystal structure.
| Color/physical appearance | metallic, silvery-white |
| Melting point/freezing point | 1297 K (1024 °C, 1875 °F) |
| Boiling point | 3347 K (3074 °C, 5565 °F) |
| Density | 7.01 g cm-3 at 20° |
| Malleability | Yes |
| Ductility | Yes |
| Electronegativity | 1.14 (Pauling Scale) |
Chemical Properties of Neodymium
- Neodymium exhibits a moderate level of reactivity. As an example, it goes through a chemical reaction with oxygen present in the air, that causes the formation of a yellowish layer. For the purpose of preventing the occurrence of tarnish, it is common to store the metal in mineral oil and encase it with a layer of plastic.
- Neodymium metal possesses a propensity for rapid oxidation. The compounds that result exhibit oxidation states of +2, +3, and +4, which correspond to pink, purple/blue, and yellow colors, respectively.
- Neodymium exhibits a high propensity for combustion at a temperature of 150°C, resulting in the formation of neodymium (III) oxide. The process of oxidation causes the spontaneous detachment of the oxide layer, which exposes the underneath layers to additional oxidation.
- Neodymium exhibits reactivity towards water, with a relatively sluggish reaction observed in the presence of cold water and a more rapid reaction observed in the presence of hot water. Therefore, it can be observed that the rate of reaction exhibits a positive correlation with the temperature of the water.
- The neodymium metal exhibits a highly vigorous reaction when combined with halogens, resulting in the formation of trihalides.
Chemical Reaction of Neodymium
- The Reaction of Neodymium With Air
Neodymium metal exhibits a gradual oxidation process when exposed to the air, leading to the formation of Neodymium (III) oxide, commonly known as Nd2O3, which immediately combusts.
4 Nd (s) + 3 O2 (g) → 2 Nd2O3 (s)
- The Reaction of Neodymium With Water
Neodymium exhibits a progressive reaction when exposed to cold water, whereas its reaction with hot water is characterized by a more expedited process. This chemical interaction leads to the production of neodymium hydroxide (Nd(OH)3) and the liberation of hydrogen gas (H2).
2 Nd (s) + 6 H2O (g) → 2 Nd(OH)3 (aq) + 3 H2 (g)
- The Reaction of Neodymium With Halogens
The element neodymium exhibits a propensity to engage in chemical reactions with various halogens, resulting in the formation of neodymium (III) halides.
The chemical reaction between neodymium metal and fluorine gas (F2) results in the formation of neodymium (III) fluoride, denoted as NdF3.
2 Nd (s) + 3 F2 (g) → 2 NdF3 (s) [violet]
The chemical reaction between neodymium metal and chlorine gas (Cl2) results in the formation of neodymium (III) chloride, denoted as NdCl3.
2 Nd (s) + 3 Cl2 (g) → 2 NdCl3 (s) [mauve]
The chemical reaction between neodymium metal and bromine (Br2) results in the formation of neodymium (III) bromide, denoted as NdBr3.
2 Nd (s) + 3 Br2 (g) → 2 NdBr3 (s) [violet]
The chemical reaction between neodymium metal and iodine represented as I2, results in the formation of neodymium (III) iodide, denoted as NdI3.
2 Nd (s) + 3 I2 (g) → 2 NdI3 (s) [green]
- The Reaction of Neodymium With Acids
The neodymium metal exhibits a high degree of solubility in dilute sulphuric acid, resulting in the formation of solutions that contain the aquated Nd(III) ion, which imparts a lilac coloration. Concurrently, the liberation of hydrogen gas, H2, takes place. The existence of Nd3+(aq) as predominantly the complex ion [Nd(OH2)9]3+ is highly probable.
2 Nd (s) + 3 H2SO4 (aq) → 2 Nd3+ (aq) + 3 SO42- (aq) + 3 H2 (g)
Uses of Neodymium
Numerous significant applications exist for neodymium and its compounds some of which are discussed here:
Used As Magnets
Permanent magnetic materials are created by combining neodymium with iron and boron. These magnets are utilized in a variety of electronic equipment, including loudspeakers (woofers), microphones, power motor generators, mobile phones, wind turbines, and electrically powered musical instruments like guitars.
Used In Laser Treatment
Neodymium is used in neodymium yttrium aluminum garnet (Nd:YAG) lasers. An apparatus that emits strongly focused light of a single hue is called a laser. The Nd: YAG laser is used to treat a few eye conditions as well as bronchial cancer. The bronchi, known as air tubes, allow air to enter the lungs.
Used In Bulbs
Neodymium finds application in diverse optical devices as well. A good example of this can be observed in a product line owned by GE Company, which features a lightbulb named the “Enrich” bulb. The glass envelope of the light bulb incorporates a minor quantity of neodymium, which serves to selectively attenuate wavelengths associated with yellow and green hues emitted by the filament.
Used In The Glass Industry
The incorporation of neodymium oxide into glass melts results in the formation of neodymium glass. The optical properties of these spectacles manifest a lavender hue when exposed to natural or incandescent illumination while exhibiting a pale blue tint when subjected to fluorescent lighting conditions.
Used In Fertilizers
Certain compounds containing rare-earth elements are utilized in the formulation of fertilizers. The utilization of rare earth element ions has been found to facilitate and enhance the growth of plants.
Used In Cryo-Coolers
The utilization of neodymium in cryocoolers is attributed to its substantial specific heat capacities at temperatures associated with liquid and helium.
Miscellaneous
The utilization of neodymium isotopes, as documented in marine sediments, serves as a valuable tool for the reconstruction of historical alterations in ocean circulation.
Neodymium salts find application in the pigmentation of glasses and enamels.
The utilization of Neodymium metal finds use in the processes of steel cutting as well as welding.
Neodymium finds application in the therapeutic management of cutaneous malignancies.
Health Effects of Neodymium
- The concentration of neodymium in the human body is relatively minute, and despite lacking a biological function, it can exert certain influences on bodily components. Notably, neodymium particulates and salts exhibit a pronounced propensity to induce irritation in ocular tissues.
- The ingestion of neodymium salts is considered to have a low level of toxicity when they are soluble, and no toxicity when they are insoluble.
- Neodymium dust particles have been observed to induce significant ocular irritation.
- In the context of occupational settings involving rare-earth elements, it is possible for individuals to inhale neodymium-based gases. Prolonged exposure to certain factors may lead to the development of pulmonary embolism.
Environmental Effects of Neodymium
- Neodymium is ubiquitously released into the environment across various locations, predominantly attributed to the activities of petroleum-based industries.
- Moreover, it is worth noting that the release of home appliances into the surrounding environment may take place in the event of their disposal.
- Neodymium will slowly build up in soils and water soils, which will lead to higher levels of the element in people, animals, and soil particles.
- The presence of neodymium in aquatic organisms has been found to induce detrimental effects on cellular membranes, thereby adversely impacting reproductive processes and compromising the functionality of the nervous system.
Video on Neodymium
References
- https://en.wikipedia.org/wiki/Neodymium_magnet
- https://www.rsc.org/periodic-table/element/60/neodymium
- https://www.britannica.com/science/neodymium
- https://www.acs.org/greenchemistry/research-innovation/endangered-elements/neodymium.html
- https://byjus.com/chemistry/neodymium/

