Mass spectroscopy is the accurate method for determining of molecular mass of the compound and its elemental composition.

Principle of Mass Spectroscopy
When molecules are bombarded with an energetic electron beam, 1e- is removed from the molecule. The removal of electron e- from a molecule causes the molecule to become positively charged, resulting in the formation of a molecular ion. Further fragmentation of molecular ions produces daughter ions.
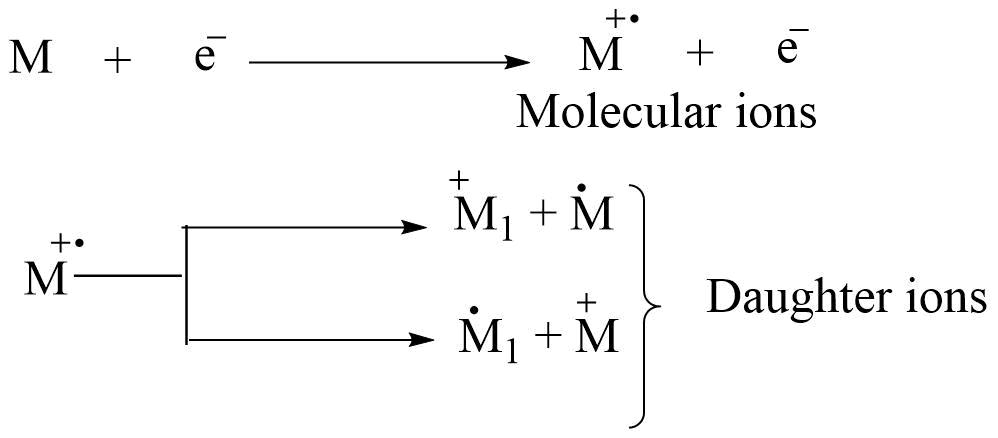
Each ion has its particular mass-to-charge ratio (i.e. m/z). They are separated according to their m/z ratio and give a mass spectrum. m/z vs abundance gives the mass spectrum.
Different terms involved in the mass spectrum
A mass spectrum is a record of the masses and relative abundances of the ions. The m/z ratios are taken along the abscissa, while the relative abundances (intensity) are taken along the ordinate.
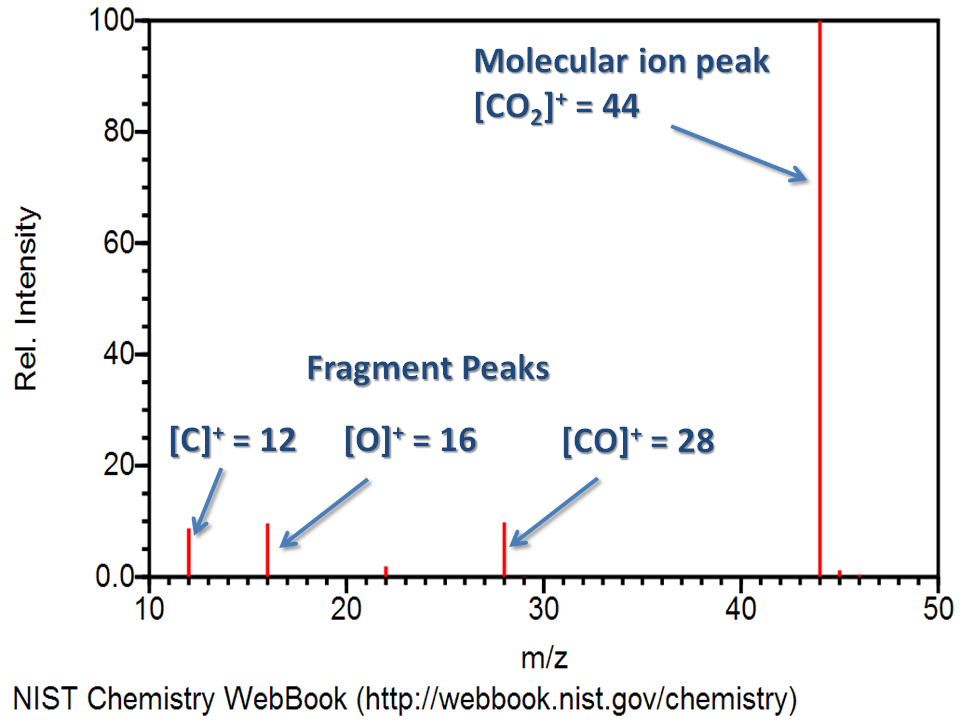
- Base peak: It is the highest peak or more intense peak in the spectrum.
- Molecular ion peak: Molecular ion is also called the parent ion and is usually designated as M+. , a positively charged molecule with an unpaired electron. The mass of the parent ion gives the molecular mass of the sample.
In a few cases, the parent ion peak may be the base peak and can be easily recognized. In most cases, the parent ion peak is not a base peak and has a very small abundance.
Some features of molecular ion peak:
- The molecular ion peak in aromatic compounds is relatively much intense due to the presence of a π-electron system.
- Conjugated olefines show a more intense molecular ion peak as compared to the corresponding non-conjugated olefins with the same number of unsaturation.
- Unsaturated compounds give a more intense peak as compared to saturated or cyclic molecules.
- The relative abundance of molecular ion peaks of straight chain compounds is more than the corresponding branched-chain compound with the same number of carbon atoms. for example, the molecular ion peak for n-pentane is more intense than that of neopentane.
- The absence of a molecular ion peak in the mass spectrum means that the compound under examination is a highly branched or tertiary alcohol.
- Fragment ion peak: The ions formed by the fragmentation of molecular ions or other ions in a mass spectrometer give a different peak in the mass spectrum. These are called fragment ion peaks. The peak of fragment ions provides information that can be used to identify unknown compounds.
- Metastable ion: Metastable ions are the ions that fragment slowly after the emergence from the ion source but before they reach the detector. They are thus displaced from the position in the mass spectrum, which would correspond to their true masses. Peak arises due to the metastable ions being normally broad and of low intensity. Such metastable peaks appear at noninteger masses.
If the ions (molecular or fragment) M+ are accelerated before their breakdown then they may decompose in the accelerating chamber into M2+ and M3, and part of the kinetic energy of M1+ is lost to the neutral fragment M3. M2+ is continuous to accelerated and collected.
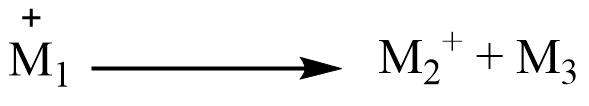
M2+ produced in this way is not recorded as mass M2 but recorded as M*,
where M*= M22/M1
M* is called metastable ions and is usually recorded as a weak broad peak of low intensity.
These metastable peaks are observed primarily when the lifetime of original species M is in the range of 10-4 – 10-6 sec, long enough to reach the accelerating region but not long enough to become fully accelerated. The recognition of metastable ions helps in the prediction of the position of parent ions and daughter ions and in determining both the original mass and size of the ejected fragment. hence it is useful for deducing the fragmentation mechanism.
Characteristics of these peaks are as follows:
- They don’t necessarily occur at the integral m/z value.
- These are much broader than the normal peaks.
- These are of relatively low abundance.
Nitrogen Rule
Nitrogen-containing compounds with an odd number of nitrogen atoms in the molecule must have an odd molecular mass. An even number of nitrogen atoms or the absence of nitrogen in the molecule show the molecular mass of the compound must be even. For the nitrogen rule to hold only unit atomic masses (i.e., integers are used in calculating the formula masses). This rule holds for all compounds containing carbon, hydrogen, oxygen, nitrogen, sulfur, and halogen, as well as many of the less usual atoms such as phosphorous, boron, and arsenic.
General fragmentation modes:
The relative abundance of the fragment ions formed depends upon the following factors:
- Stability of ions
- Stability of radical lost
the stability of the ions can be judged by stabilization of the charge, which depends upon;
a. Resonance
b. Inductive effect
c. Polarisability and so on
Some important fragmentation mods are:
Simple cleavage
Fragmentation of odd electron molecular ion M+. may occur through homolytic or heterolytic cleavage of single bond.
A. Homolytic cleavage:
Odd electron ions have an unpaired electron which is capable of new bond formation. The energy released by bond formation can help offset the energy required for the cleavage of some other bond in the molecules.
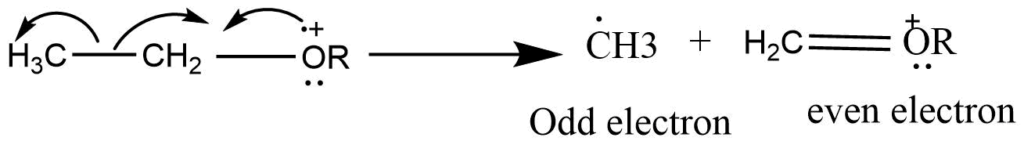
Homolytic cleavage reactions are very common and can be classified in the following steps:
Mode I: This type of fragmentation mode operates in compounds in which the heteroatom is singly bonded to a carbon atom. In this mode, the parent ions are formed by the removal of one electron from the heteroatom. A new bond is formed with an adjacent atom through the donation of an unpaired electron and the transfer of an electron from the adjacent bond.

More abundant peaks are formed by the cleavage of the C-C bond, which is in the α position to the heteroatom in mass spectra of alcohol, amines, and ethers.
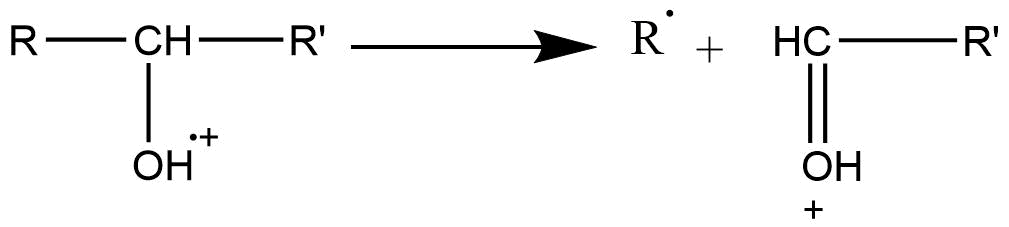
Mode II: when a heteroatom is bonded to the carbon atom by the double bond, α-cleavage is the preferred fragmentation mode.
This type of fragmentation is shown by many compounds such as ketone, and ester amide. In ketones, significant peaks are obtained due to cleavage of the C-C bond, which is alpha to the carbonyl bond. Unsymmetrical ketones show two types of peaks since either alkyl group can lose. The higher alkyl radical is usually preferred.
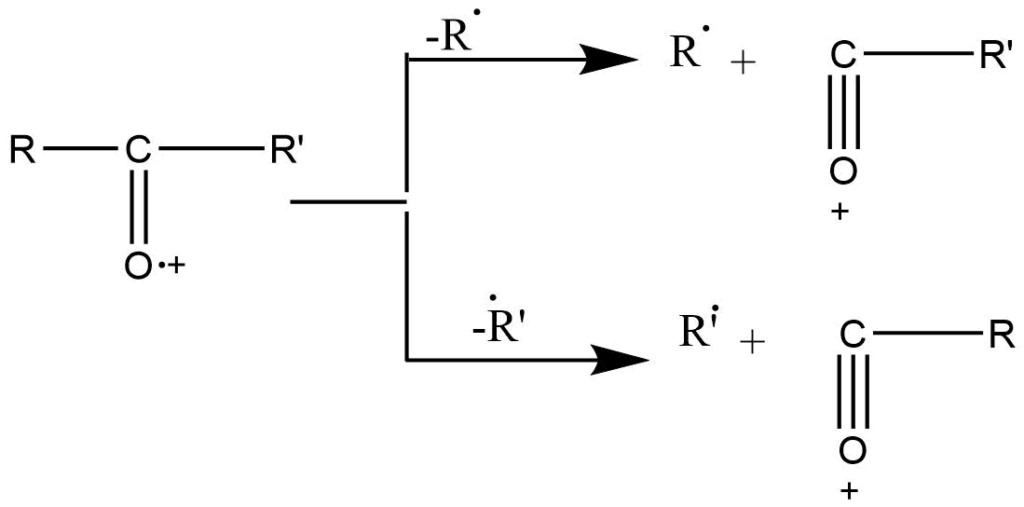
B. Heterolytic cleavage
Cleavage of the C-X (x= O, N, S, Cl) bond is more difficult than that of the C-C bond. In such a cleavage the positive charge is carried by the carbon atom and not by a heteroatom. eg. Fragmentation of alkyl halide.
In the spectra of monohalogenated compounds, hydrocarbon ions are formed in more abundance. As the size of the halogen atom increases, the C-X bond becomes weak, so alkyl bromide and iodides are easily broken, while alkyl chloride is less susceptible to fragmentation.
Retro-Diel’s Alder reaction
It is the characteristic of cyclic olefins, which involve multicentered fragmentation. It involves the cleavage of two bonds of the cyclic system resulting in the formation of two stable unsaturated fragments in which two new binds are formed. This process is not accompanied by any hydrogen transfer rearrangements.
The highly substituted or more conjugated fragment, which has low ionization energy, carries a charge.
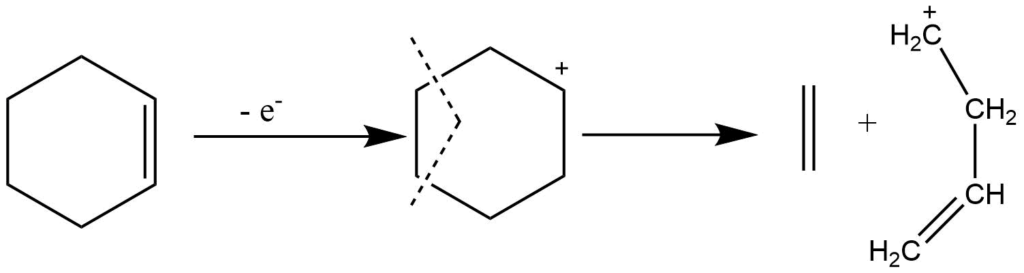
Rearrangement reactions
Rearrangement ions are fragments whose origin can’t be described by simple cleavage of bond in the molecular ion but are a result of intramolecular atomic rearrangement during fragmentation.
- Rearrangement resulting in the elimination of stable neutral molecules is common.
- The rearrangement peak can be recognized by considering the m/z number of fragments ions and their corresponding molecular ion.
- An even-numbered peak derived from even-numbered molecular ions is the result of two cleavages, which may involve a rearrangement.
Different modes of rearrangements
A. Hydrogen transfer
These processes are very common in mass spectroscopy, which involves the transfer of hydrogen atoms from one part of the molecule to another part. E.g., an even-numbered fragment mass from an even-numbered molecular ion mass indicates a rearrangement of hydrogen has accompanied the fragmentation process.
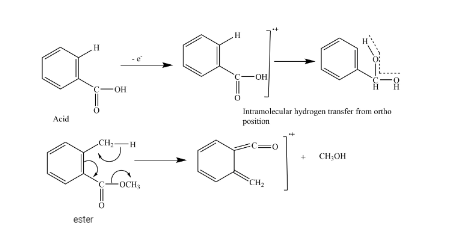
From hydrogen rearrangement, we can distinguish between cis and trans isomer as well as ortho-substituted compounds from meta and para-substituted compounds.
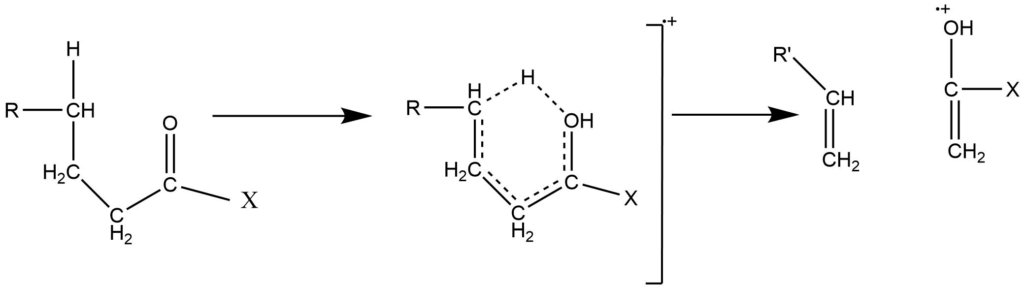
B. γ -hydrogen transfer (Mc-Lafferty rearrangement)
This arrangement involves the cleavage of the β-bond followed by the γ-Hydrogen transfer. The mechanism involves the six-membered transition state. This rearrangement eliminates neutral molecules from aldehydes, ketones, amines, and unsaturated compounds.
To undergo the Mc-Lafferty rearrangement reaction, a molecule must possess appropriately located heteroatoms (e.g., oxygen), a π system, and an abstractable hydrogen atom at γ position to the C=O system.
such arrangement often accounts for prominent characteristic peaks and is consequently very useful. They can frequently be rationalized on the basis of low energy transitions and increased stability of the product.
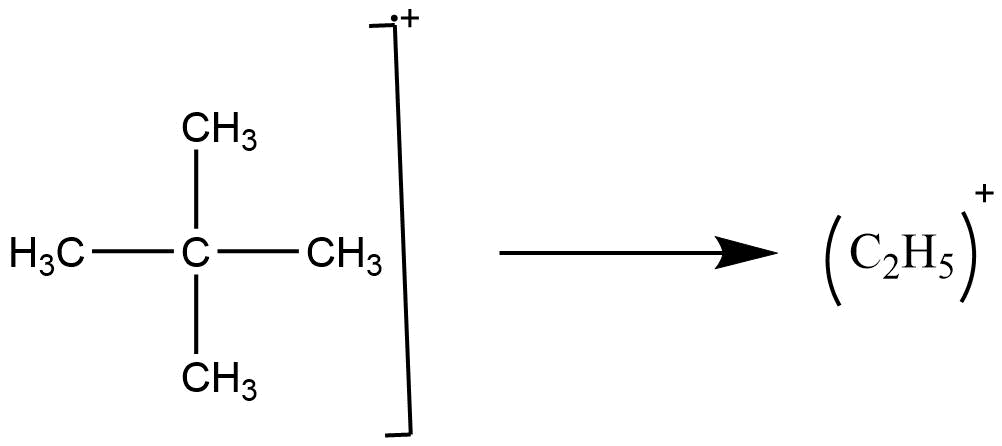
C. Random rearrangement: Random rearrangement of hydrocarbon was noted by early mass spectrometers in the petroleum industry.
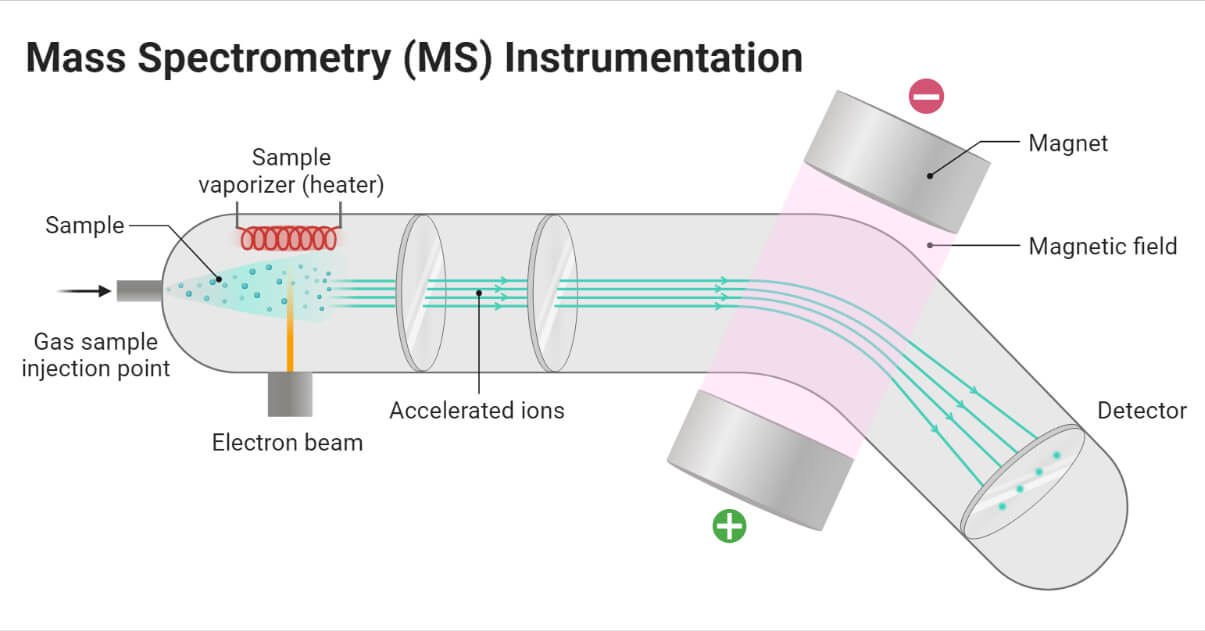
The mass spectrometer is generally designed to perform three basic functions, they are:
- To vaporize the compounds of various volatility.
- To produce ions from the neutral compounds in the vapor phase.
- To separate the compounds according to their mass-to-charge ratio and record them.
Instrumentation of Mass Spectrometry (MS)
Sample handling system
The sample handling system is used to introduce the sample into the ion source at low pressure. Heating is required for less volatile sample. Solid, liquid, and gas samples can be used in mass spectrometry.
Ionization process in Mass Spectrometry
Samples are ionized by removing one or more electrons to give the molecular ions and fragment ions. Different ionization methods are used in mass spectroscopy. They are
Electron ionization/Electron impact
This is the oldest and best-characterized method of ionization. In this process, the beam of electrons passes through the gas sample. The electrons collide with the neutral molecule and knock off another electron to form molecular ions or fragment ions.
Chemical ionization
The chemical ionization process involves reagent gas like methane, isobutane or ammonia. First, the reagent gas is ionized by electron impact, and the reaction between reagent gas ions and the sample produces the ions of the analyte.
Field desorption
In this process, the sample is placed on the anode of pair of electrodes, and the electrodes are placed at too high a potential. SO, the sample desorption occurs to produce molecular and quasimolecular ions with sufficient internal energy for extensive fragmentation.tt
Atmospheric pressure chemical ionization (APCI)
In APCI, a corona discharge is used to ionize the sample in the atmospheric region. The gas phase ionization in APCI is more effective for analyzing less-polar species.
Fast atom bombardment (FAB)
It involves the bombarding of samples by using beams of xenon atoms of high transitional energy. In this process, xenon atoms are first ionized by using the electrons to give xenon radical cations. The xenon radical cations are accelerated to 6-10 keV to give radical cations of high transitional energy. The radical cations are then passed through xenon to form the high-energy xenon atom Xe and Xe.+ions are removed by the electric field.
The compounds of interest are placed in a metal sheet and ionized by a high-energy beam of a xenon atom.
Electrospray ionization
The electrospray ionization involves placing the ionizing voltage across the nebulizer needle present at the outlet from high-performance liquid chromatography. ESI can produce the multiply charged ions with the number of charged tending to increase with an increase in molecular weight. This is popularly used for water-soluble biomolecules like protein, peptides, and carbohydrates.
Matrix-assisted laser desorption ionization
In this process, the sample is dissolved in the solution containing an excess of a matrix like dihydroxy benzoic acid, and sinapic acid, which consists of chromophores, capable of absorbing laser light. When the solution is placed in the laser light the solution absorbs the laser light and produces plasma, which results in ionization and vaporization of the sample.
Acceleration of ions
Ions are accelerated to attain the same kinetic energy. The ions pass through three accelerating slits with decreasing voltage.
Deflection of ions
Ions are deflected by using the magnetic field on the basis of their m/z ratio. The lighter ions with a high positive charge are more deflected than others.
Detection of ions
Detectors detect the ions passing through the mass analyzer on the basis of the m/z ratio.
Mass analyzers of Mass Spectrometry
Different mass analyzers are:
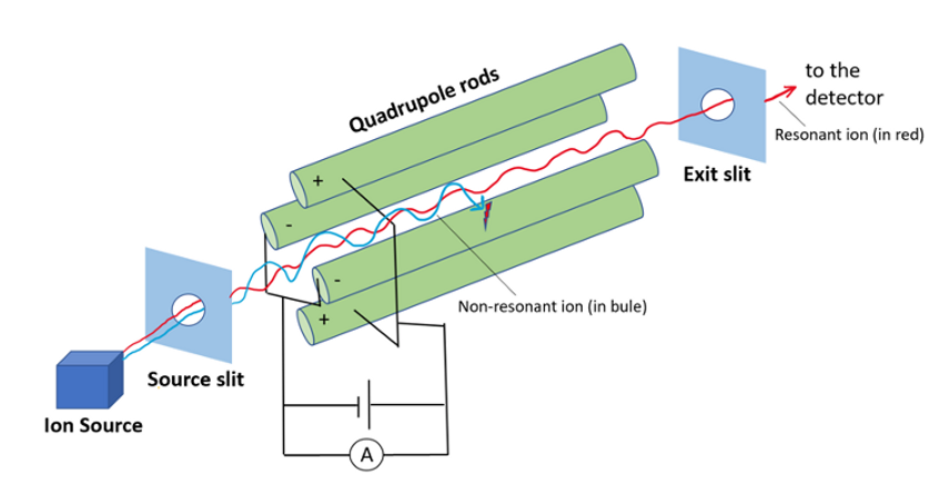
Quadrupole mass filter
Qurdupole mass filter consists of four cylindrical rods arranged in squares parallel to the direction of the ion beams. In this radiofrequency and direct current are applied. The combination of Rf and direct current generates the oscillating electrostatic field between the region of ions. When the ions enter into the mass analyzer depending upon the ratio of Rf and Dc voltage oscillating electrostatic field will be generated for ions. Ions with an inappropriate m/z ratio will undergo an unstable oscillation and hit the rods. But ions with the correct m/z ratio undergo stable oscillation and strike the detector to give a signal.
Quadruple ion trap:
It is the spherical configuration of a linear mass filter. The difference between a linear mass filter and an ion trap is that the linear mass filter directs sorted ions through the detector, whereas the ion trap temporarily retains unsorted ions within the trap. After scanning the electric field, they are released to the detector. Because of their speed and sensitivity, quadrupole ion trap methods are well suited for use with capillary gas chromatography. These are compact, inexpensive, and easy to use.
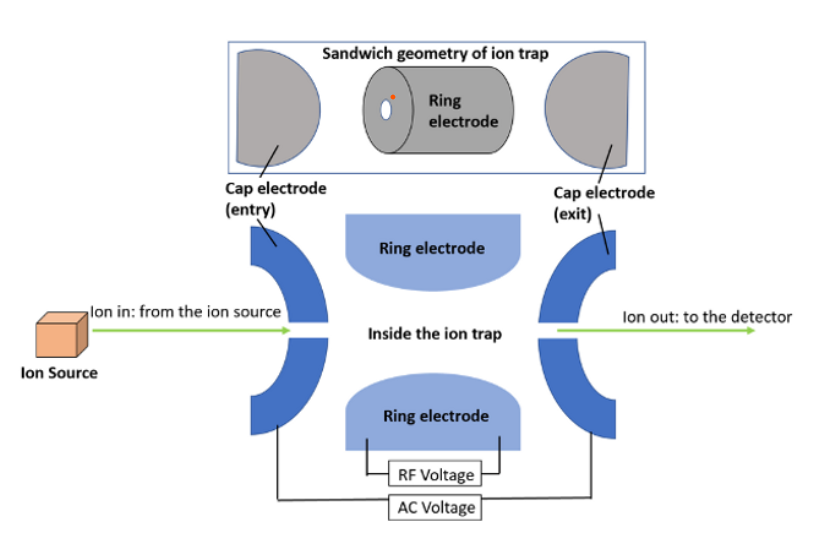
Time of flight mass spectrometer
In this procedure, ions are subjected to a high potential difference (V) to attain the same transitional energy in electron volt. They are further accelerated and transferred to the field-free region. There they are separated overtime on the basis of their m/z values. This is simply based on the principle that the velocities of two ions depend on the mass of ions. In this process, ions should be created at the same instant and they should have the same kinetic energy. The lighter ions have higher velocity compared to the higher ions, so on passing towards the detector, they hit the detectors first.
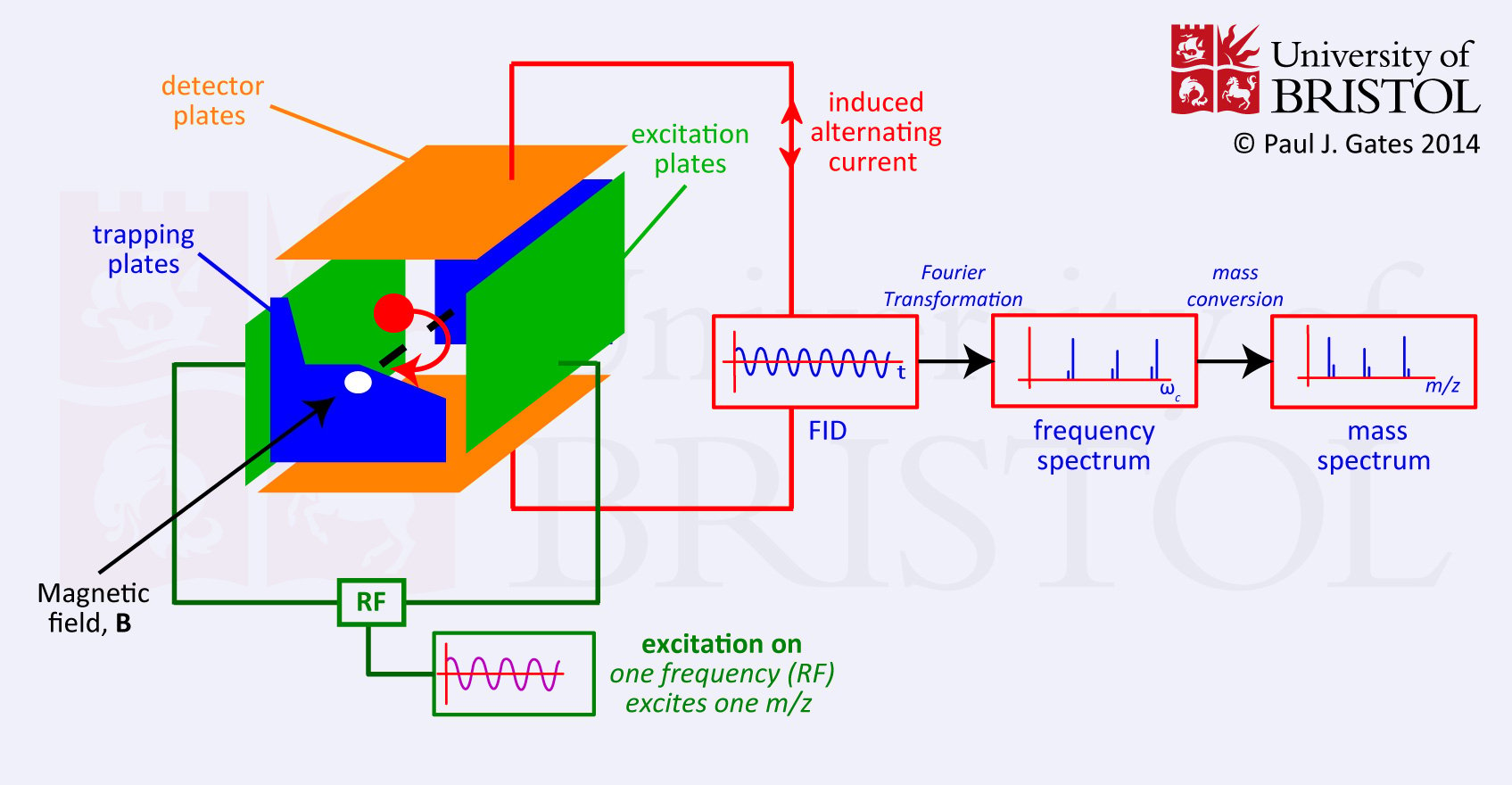
Fourier transform-Ion Cyclotron Resonance
Ions, generated by the electron beam from heated filament are passed into cubic cells under vacuum conditions. An electric tapping potential and a magnetic field hold these ions in place. Depending on their m/z ratio, each ion assumes the cycloidal orbit at its own characteristic frequency. By varying the electric field, these frequencies are scanned until the cycloidal frequency is in resonance with the applied radio frequency. The motion of ions with the same frequency is coherent at resonance, and a signal is detected.
Detectors of Mass Spectrometry
Photoplate detectors
It is the earliest and the largest ion detector. It involves the dispersion of ions along the lengthy focal plane resulting in adequate unit mass resolution across the entire spectral region. So photo plate detector makes able to have simultaneous detection and signal integration.
Electron multipliers
The ions emerging from the mass analyzer are detected using an electron multiplier. It is a common type of ion detector that comes in a variety of designs. In this detector, ions striking the conversion dynode generate secondary ions. The dynodes then generate the electron cascade, effectively multiplying the single incident ions by 106 or more. The degree of multiplication is determined by the individual dynode surface composition, acceleration per stage (bias voltage), the number of dynodes (6-20), and bias current circuit design.
Photomultiplier
Ions strike the photomultiplier detector, resulting in electron emission. The emitted electron then strikes the phosphor screen, giving rise to photons. After that, the photons are amplified by passing through the multiplier. The advantage of this detector is that it operates in a vacuum state, which prevents contamination and extends the detector’s lifetime.
Faraday cups
The name faraday cup is named after Michael Faraday. In this detector, a metal cup is placed in the path of the ion beam and is connected to an electrometer that measures the current of the ion beam. When the incident ions strike the dynode surface, it produces electrons and induces a current, which is amplified and recorded. Faraday cups can detect the higher current, which cannot be measured by an electron multiplier.
Photographic plates
These are the time integrating devices generally used with the radiofrequency spark instruments. Photographic plates integrate the ion signal over time and detect the ions of all masses simultaneously. It is the simplest and oldest type of ion detector.
For more details on the instrumentation of mass spectroscopy, you can watch this video.
Application of Mass Spectroscopy
- It is used to determine the molecular weight of compounds.
- In organic chemistry, mass spectroscopy is used to determine the structure of complicated compounds.
- Mass spectroscopy is used for the qualitative and quantitative study of compounds.
- It is also used for the detection of impurities.
- Mass spectroscopy is used in the study of drug metabolism study, and also has clinical, toxicological, and forensic applications.
- It is used in the isotope ratio determination and carbon dating process.
References
- Silverstein, R. M., Webster, F. X., & Kiemle, D. J. (2005). Spectrometric identification of organic compounds. Hoboken, NJ: John Wiley & Sons.
- Analytical_Chemistry/Supplemental_Modules_(Analytical_Chemistry)/Instrumental_Analysis/Mass_Spectrometry/Mass_Spectrometers_(Instrumentation)/Mass_Analyzers_(Mass_Spectrometry)
- http://www.premierbiosoft.com/tech_notes/mass-spectrometry.html
- https://barclayphysics.fandom.com/wiki/How_are_the_ions_produced_and_detected
- https://byjus.com/chemistry/mass-spectrometry/
- https://microbenotes.com/mass-spectrometry-ms-principle-working-instrumentation-steps-applications/
- https://pubs.acs.org/doi/pdf/10.1021/ac053495p
- https://www.britannica.com/science/mass-spectrometry/Hydrogen-carbon-nitrogen-oxygen-and-sulfur-in-nature
- https://www.chemguide.co.uk/analysis/masspec/howitworks.html
- https://www.priyamstudycentre.com/2022/02/mass-spectrometry.html
- https://www.researchgate.net/publication/328661548_Mass_Spectrometry_Detectors_Review
- https://www.technologynetworks.com/analysis/articles/types-of-ion-detector-for-mass-spectrometry-347890
- https://www.technologynetworks.com/analysis/lists/eight-emerging-applications-of-mass-spectrometry-288234
- https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/massspec/masspec1.htm

