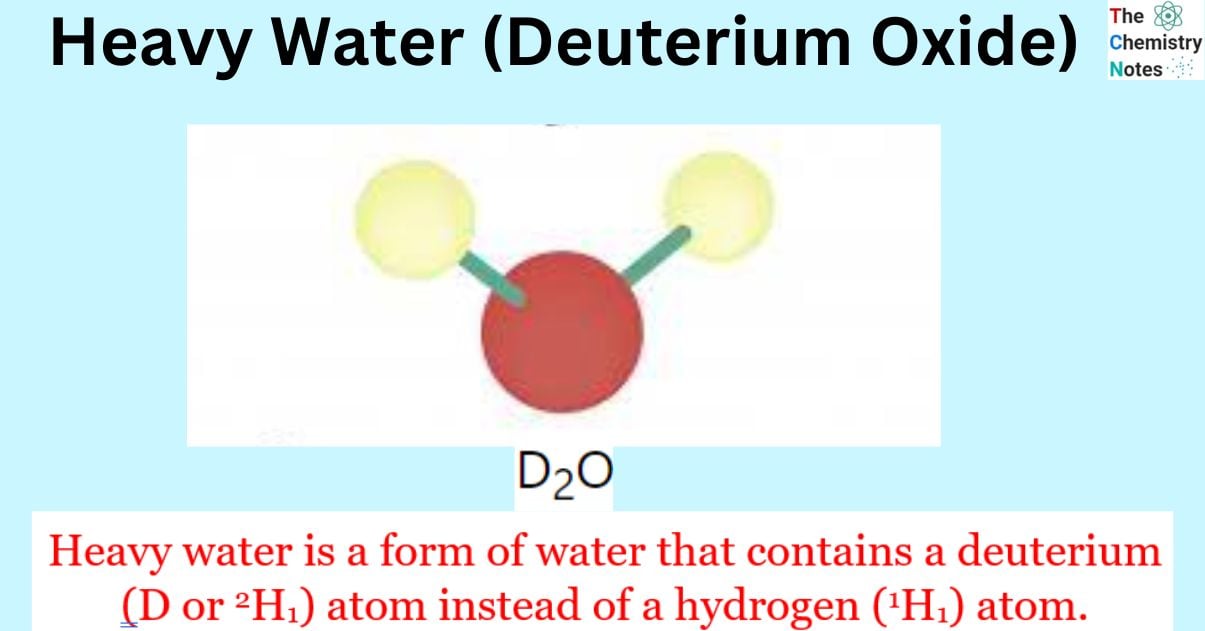
Heavy water, scientifically called deuterium oxide, is a variant of water wherein the regular hydrogen atoms are replaced by heavy hydrogen, commonly known as deuterium. Alternatively, it can be expressed as 2H2O or D2O.
Deuterium exhibits distinct characteristics compared to the hydrogen isotope frequently encountered in water and is known by the term protium. This distinction is due to the fact that every deuterium atom includes a proton and a neutron, whereas the more prevalent hydrogen isotope solely comprises a proton. The molecular mass (molar mass) of deuterium oxide is higher than that of regular water due to deuterium’s greater atomic mass than protium. The presence of deuterium in heavy water (D2O) results in slight variations in its chemical and physical characteristics in comparison to regular water (H2O).
Discovery of Heavy Water (D20)
In 1931, Harold Urey, an esteemed American scientist, and recipient of the Nobel Prize, made the groundbreaking discovery of the isotope deuterium. Subsequently, Urey successfully achieved the concentration of deuterium within water.
In 1933, Gilbert Newton Lewis, who served as Urey’s mentor, successfully obtained the initial sample of pure heavy water through the process of electrolysis.
In 1934, George de Hevesy and Erich Hofer conducted a pioneering biological tracer experiment involving the utilization of heavy water. Their objective was to estimate the rate of water turnover within the human body.
In 1934, the auto dissociation of heavy water was investigated by Emilian Bratu and Otto Redlich.
Occurrence of Heavy Water
The production of heavy water occurs through the substitution of two hydrogen atoms in regular water with deuterium atoms. Deuterium oxide, also known as D2O, is a chemical compound commonly referred to as heavy water.
Approximately 1 part of heavy water is present in every 6000 parts of regular water. Lewis and Donald successfully employed constant the electrolysis process to separate a small volume of pure heavy water from a solution including a minute amount of alkali. The remnants remaining after the snow melts in the Himalayas yielded a discovery. Trace amounts can also be discovered on the leaves of Banyan trees and in precipitation.
Preparation of Heavy Water
Multi-stage Electrolysis
Currently, the production of heavy water includes a method of extending the electrolysis process of water that contains an alkali substance, specifically a solution of sodium hydroxide (N/2 NaOH).
When the process of electrolysis takes place on the water over and over, up to the seventh iteration, approximately 99 percent of deuterium (D2) is produced.
The liberation of protium occurs readily in the context of H2O due to the higher mobility of H+ ions relative to D+ ions. This factor can be attributed to its relatively smaller dimensions. Additionally, they exhibit a reduced discharge potential, resulting in a higher propensity for H+ ions to be discharged at the cathode. Furthermore, it is observed that hydrogen atoms exhibit a greater propensity to form molecular hydrogen compared to deuterium atoms.
The electrolytic cell for the production of heavy water was designed by Brown, Degget, and Urey. The structure comprises a cylindrical steel enclosure measuring 45 cm in length and 10 cm in diameter. This object can be identified as the cathode. A significant quantity of these cells is employed in various stages of water electrolysis.
Using Fractional Distillation
Additionally, it is possible to produce it through the process of fractional distillation, in which the lighter fraction (H2O) is distilled first, resulting in a residue that is more concentrated in D2O.
A slight disparity exists in the boiling points between regular water and heavy water. The utilization of fractional distillation serves as the fundamental approach for the production of heavy water. The boiling point of regular water is 373K, while heavy water has a boiling point of 374.42K under normal atmospheric pressure.
Using Exchange Reactions
Exchange operations have the capability to generate heavy water, scientifically known as deuterium oxide (D2O). The hydrogen atoms within H2S gas can be substituted with deuterium atoms derived from D2O found in the water as it undergoes heating processes. Consequently, D2S is introduced to H2S. When the D2S-enriched H2S is introduced to cold water, a process occurs where the deuterium from the D2S and the hydrogen from the H2O undergo an exchange, resulting in the swapping of positions. The technique is iteratively applied, wherein the concentration of D2O in the cold water is progressively augmented.
Types of Heavy Water
Oxygenated Water
The distinguishing feature of heavy-oxygen water is the presence of isotopes 17O and 18O. The term “heavy water” is derived from its elevated density relative to that of ordinary water. The existence of water enriched with heavy oxygen, particularly the isotope 18O, plays a vital role in the production of the fluorine isotope 18F. Furthermore, it finds application in the fields of radiopharmaceuticals and radiotracers. Moreover, it is employed as a constituent in radiotracers and radiopharmaceuticals.
Semi-Heavy Water
Water that possesses a moderate level of density is commonly known as semi-heavy water, and it is scientifically represented as HDO. The composition consists of a single protium atom, a single deuterium atom, and one oxygen atom. Hydrogen atoms demonstrate a tendency to engage in intermolecular exchange within water molecules. This suggests that the presence of protium and deuterium, which in samples of water facilitates the detection of HDO.
Titrated Water (Super-Heavy Water)
Tritiated Water (T2O) is a variant of water that exhibits radioactivity due to the presence of tritium, a hydrogen isotope, in place of the more common protium isotope. It is also referred to as super-heavy water. The quantification of the overall water content within an organism can be achieved through the utilization of tritiated water. The compound T2O possesses a molar mass of 22.03 g mol-1 and a density of 1.85 g mL-1.
Physical Properties of Water
Heavy water is a mobile liquid that lacks color, odor, and taste. The majority of physical constants, such as melting point, boiling point, specific gravity, viscosity, specific heat, and dielectric constant, exhibit higher magnitudes compared to the corresponding values observed for standard water.
| Property | Value for H20 | Value for D2O |
|---|---|---|
| Freezing Point | 0℃ | 3.8℃ |
| Boiling Point | 100℃ | 101.42℃ |
| Density at 20℃ | 0.9982 | 1.1058 |
| Temperature of maximum density | 4.08℃ | 11.22℃ |
| Specific Heat | 1.000 | 1.018 |
| Dielectric Constant 0℃ | 81.5 | 80.7 |
| Refractive Index | 1.3329 | 1.3281 |
| Surface Tension | 72.75 dyne cm-1 | 67.2 dyne cm-1 |
| Viscosity at 20℃ | 1.009 cp | 1.26 cp |
| Solubility of NaCl at 20℃ | 35.9 g/100mL | 30.5 g/100mL |
| Ionization Constant (25℃) | 1 x 1-14 | 0.2x 10-14 |
Chemical Properties of Heavy Water
- The reactions involving deuterated water (D2O) exhibit a reduced reaction rate in comparison to those involving regular water (H2O).
- Deuterium can be obtained at the cathode through the electrolysis of heavy water.
- The chemical properties of hydrogen isotopes differ as a result of their distinct atomic masses.
- The concentration of D+ ions in D2O is typically lower than the concentration of H+ ions in H2O at a given temperature.
- The reduced reactivity of deuterium oxide (D2O) in comparison to water (H2O) can be explained by the higher dissociation energy of the O-D bond in D2O relative to the O-H bond in H2O.
Chemical Reactions of Heavy Water
Deuterolysis: Deuterolysis refers to the hydrolysis of specific inorganic salts in the presence of water. Analogous phenomena, referred to as salt Deuterolysis, are observed when heavy water is employed.
AlCl3 + 3 D2O → Al(OD)3 + 3 DCl
BaS + 2 D2O → Ba(OD)2 + D2S
Reaction on Metals: The process of heavy water reacting with highly reactive metals such as sodium and calcium results in the release of deuterium and the formation of heavy alkalis.
2 Na + 2 D2O → 2 NaOD + D2
Reaction on Metal Oxides: When basic oxides like sodium monoxide and calcium oxide react with heavy water, heavy alkalis are generated.
Na2O + D2O → 2 NaOD
Reaction on Non-Metal Oxides: Deutero acids are generated through the reaction between heavy water and acidic non-metallic oxides, including but not limited to sulfur trioxide and dinitrogen pentoxide.
SO3 + D2O → D2SO4
The Reaction of Metal Nitrides, Phosphides, and Carbides: The reaction involving metal nitrides, phosphides, and carbides is characterized by the liberation of heavy ammonia, which is when heavy water is introduced.
Mg3N2 + 6 D2O → 3 Mg(OD)2 + 2 ND3
When metal phosphides are utilized, it produces deuteron phosphine.
Ca3P2 + 6 D2O → 3 Ca(OD)2 + 2 PD3
When metal carbides are used, either deuteron acetylene or deuteron methane is generated.
CaC2 + 2 D2O → 3 Ca(OD)2 + C2D2
Formation of Salt Deuterates: Salt deuterates are made when heavy water reacts with salt. When salt dissolves in heavy water and then crystallizes, these deuterates are made. Example: CuSO4.5D2O, MgSO4.7D2O.
Exchange Reactions: Heavy water exhibits the ability to interact with a diverse range of substances that possess hydrogen atoms that are susceptible to change. As a consequence of these interactions, the hydrogen atoms may undergo either partial or complete substitution with deuterium. If the compounds contain hydrogen atoms that exhibit ionic (polar) characteristics, the likelihood of exchange processes taking place is increased.
NaOH + D2O → NaOD + HDO
HCl + D2O → DCl + HDO
Biological Properties of Heavy-Water
Heavy water is hazardous to people, plants, and animals because it reduces various reactions, which in turn slows down the rate at which those reactions occur to them. Both plants and animals grow more slowly in muddy water. In pure, heavy water, seeds do not germinate, and small fish die. In addition, water that contains a lot of D2O is poisonous.
Uses of Heavy Water
Heavy water can be used for various applications some of which are discussed here:
Nuclear Magnetic Resonance
Nuclear Magnetic Resonance (NMR) spectroscopy employs deuterium oxide (D2O) as a solvent to mitigate the interference caused by the interaction between the signal that is emanating from H2O solvent molecules and the signal originating from the molecule under analysis. Deuterium oxide (D2O) exhibits a distinct magnetic moment, rendering it advantageous for utilization in nuclear magnetic resonance spectroscopy.
Neutron Moderator
The utilization of heavier water (D2O) as a neutron moderator is observed in specific categories of nuclear reactors. The process of neutron moderation facilitates the reduction of neutron velocity, thereby enhancing the likelihood of their interaction with fissile uranium-235 rather than uranium-238. Consequently, this process aids in the capture of neutrons without undergoing fission.
In Organic Chemistry
In the field of Organic Chemistry, the synthesis of specifically labeled isotopologues of organic compounds necessitates the utilization of deuterium. Deuterium oxide (D2O) is frequently employed as a primary source of deuterium.
Metabolic Rate Testing
The evaluation of the mean rate of metabolism in both humans and animals necessitates the utilization of heavy water together with H218O as a component of the testing procedure in the fields of physiology and biology.
Tritium Production
Tritium, the primary constituent employed in controlled nuclear fusion reactions, is generated through the process of capturing neutrons by deuterium, which is found in heavy water.
Frequently Asked Questions (FAQ)
Can you drink heavy water?
The body won’t be harmed if you consume a tiny amount of heavy water.
Why is heavy water a better moderator than light water?
Heavy water is a better moderator than light water because, compared to light water, it does not capture thermal neutrons.
Whose polarity is greater, D2O or H2O?
Deuterium is more electronegative than hydrogen, and as the electronegative differences between Deuterium and oxygen widen, so does the dipole moment of D2O. D2O is therefore more polar than H2O.
What is the nature of D2O? Acidic or basic?
The pH at room temperature obtained for heavy water D2O is greater than that of normal water. pH of D2O is around 7.44, which indicates that D2O is basic in nature.
What exactly is a Moderator?
In a nuclear reactor, a moderator is necessary to slow down the neutrons produced during the fission event so that the chain reaction can continue. Because of its high moderating ratio and low neutron absorption cross-section, heavy water is an effective moderator.
How much will heavy water harm you?
If 50% of the usual water in the human body is replaced with heavy water, it might be lethal
What is Deuterium Depleted Water?
Deuterium Depleted Water is water that contains less Deuterium than its natural abundance (125 ppm). It is becoming more popular as an adjuvant therapy for cancer.
Depleted Deuterium Water contains less deuterium than natural water (125 ppm). It is becoming more popular as an adjuvant cancer treatment.
Video on Heavy Water
References
- https://www.geeksforgeeks.org/heavy-water-definition-occurrence-preparation-properties/
- https://unacademy.com/content/neet-ug/study-material/chemistry/properties-of-heavy-water/
- https://www.toppr.com/ask/en-np/content/concept/heavy-water-preparation-properties-and-uses-203930/
- https://www.britannica.com/science/heavy-water
- https://www.chemistrylearner.com/heavy-water.html
- https://www.aakash.ac.in/important-concepts/chemistry/heavy-water
- Isotopic Effects of Heavy Water in Biological Objects Oleg Mosin, Ignat Ignatov
- Thomson, J.F. (1960). “Physiological Effects of D2O in Mammals. Deuterium Isotope Effects in Chemistry and Biology”. Annals of the New York Academy of Sciences. 84 (16): 736–744. Bibcode:1960NYASA..84..736T. doi:10.1111/j.1749-6632.1960.tb39105.x

