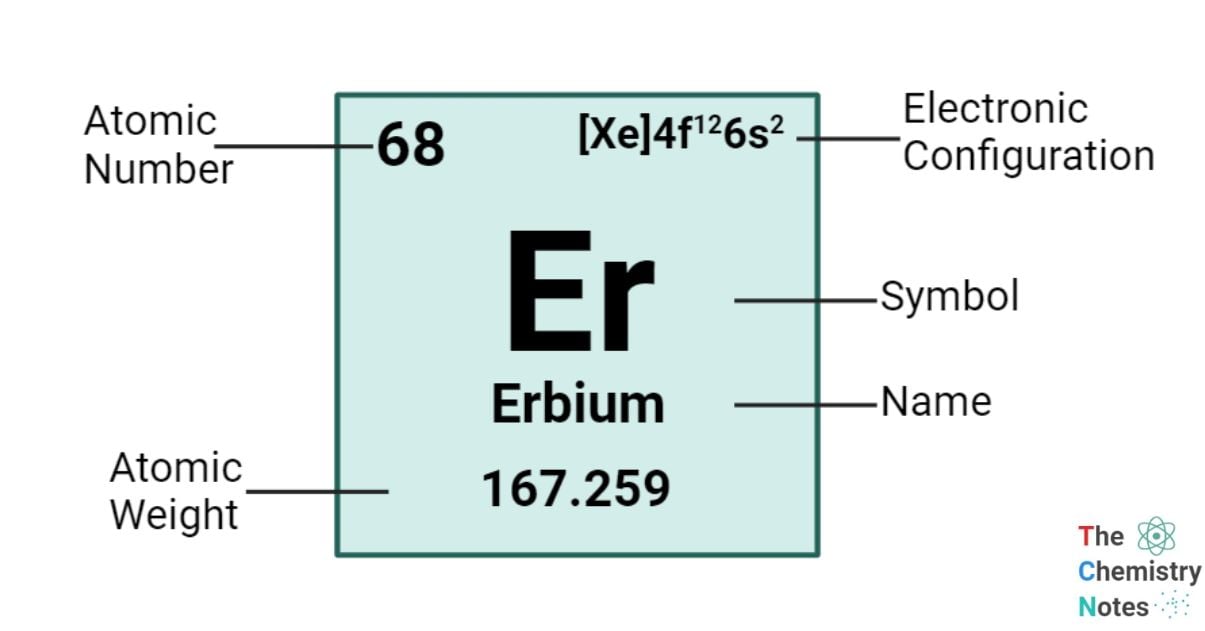
Erbium is a chemical element with an atomic number of 68 and is represented by the symbol ‘Er’ in the periodic table. It is hard and silvery in appearance and classified as rare earth metal and belongs to the f-block of the lanthanide group of the periodic table. Erbium, similar to the majority of rare-earth elements, is frequently encountered in its +3 oxidation state. However, it is worth noting that erbium exhibits additional oxidation states, specifically 0, +1, and +2.
Erbium is found to be the 44th most prevalent element within the Earth’s crust. Its abundance is estimated to range from approximately 3.0 to 3.8 parts per million (ppm). Similar to other rare earth elements, this particular element is not naturally encountered in its elemental form, but rather exists in a state of bounds within monazite sand ores, bastnasite, xenotime, and euxenite.
Interesting Science Videos
History of Erbium
- In the year 1843, the distinguished Swedish chemist Carl Gustaf Mosander successfully conducted a process of isolation on gadolinite, a mineral that had previously been unearthed in a quarry situated within proximity to Ytterby. This procedure resulted in the production of three distinct substances, which Mosander subsequently designated as yttria, erbia, and terbia.
- The researcher successfully synthesized terbium and erbium by subjecting terbia and erbia to a chemical process. Subsequent investigations revealed that terbia corresponded to terbium oxide, exhibiting a yellow hue, while erbia denoted erbium oxide, characterized by a pink coloration.
- Nevertheless, it is worth noting that the initial specimens of the aforementioned metal comprised additional elements from the group of rare earth elements. In the year 1934, the esteemed scientists Wilhelm Klemm and Heinrich Bommer successfully generated a pristine sample through the process of heating meticulously purified erbium chloride and potassium.
Occurrence of Erbium
- Erbium is identified as the 44th most abundant element in the Earth’s crust. The predicted abundance of the subject is believed to fall within the range of approximately 3.0 to 3.8 parts per million (ppm).
- Like other rare earth elements, this specific element is not naturally found in its elemental state, instead, it is present in its bound composed within monazite sand ores, bastnasite, xenotime, and euxenite.
- The industrial manufacturing of this substance entails a highly advanced ion exchange procedure, whereby xenotime and euxenite minerals are subjected to refinement to acquire it.
- The process of isolating erbium from its oxide or salts entails subjecting the compound to a significantly elevated temperature of 1450°C in the presence of calcium under an argon atmosphere.
- Er exhibits a diverse range of isotopes, totaling 31 in number, each possessing distinct half-lives. These isotopes span a mass number spectrum from 145 to 175. Erbium found in nature is composed of a combination of six distinct isotopes.
Isotopes of Erbium
Er consists of six naturally occurring isotopes that include: 162Er, 164Er, 166Er, 167Er, 168Er, and 170Er.
Naturally Occurring Isotopes of Erbium
| Isotopes | Natural Abundance (% atoms) |
|---|---|
| 162Er | 0.14 |
| 164Er | 1.61 |
| 166Er | 33.61 |
| 167Er | 22.93 |
| 168Er | 26.78 |
| 170Er | 14.93 |
Elemental Properties of Erbium
| Electronic Configuration | [Xe] 4f12 6s2 |
| Atomic Number | 68 |
| Atomic Weight | 167.26 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | Lanthanides, 6, f-block |
| Density | 9.066 g/cm3 at 20 °C |
| Appearance | silvery-white |
| Van der Waals radius | unknown |
| Electron shells | 2, 8, 18, 30, 8, 2 |
| Electrons | 68 |
| Protons | 68 |
| Neutrons in the most abundant isotope | 98 |
Physical Properties of Erbium
- Er has an atomic number of 68 and is a silvery-white rare earth metal. It has a melting point of 1529 °C (2784 °F) and a boiling point of 2868 °C (5194 °F).
- Er has a solid phase density of 9.066 g/cm3 and a liquid or molten phase density of 8.86 g/cm3.
- It is malleable which means it can be easily beaten into thin sheets without any cleavage.
- It is ductile which means it is possible to draw thin wires from it without breaking.
- Erbium oxide exhibits a distinctive pink hue, while erbium salts manifest a captivating rose-pink coloration.
- Er exhibits a notable degree of stability in atmospheric conditions, demonstrating a comparatively slower rate of oxidation when compared to other elements within the lanthanide series.
- Er exhibits ferromagnetic properties at temperatures below 19 Kelvin (-254.15 °C). Within the temperature range of 19-80 Kelvin (-254.15 to -193.15 °C), the material exhibits antiferromagnetic properties. At temperatures exceeding 80 Kelvin (-193.15 °C), the material exhibits paramagnetic ordering.
- The properties of Er are significantly influenced by the impurities that are present within it.

| Color/physical appearance | metallic, silvery-white |
| Melting point/freezing point | 1802 K (1529 °C, 2784 °F) |
| Boiling point | 3141 K (2868 °C, 5194 °F) |
| Density | 9.066 g cm-3 at 20° |
| Malleability | Yes |
| Ductility | Yes |
| Electronegativity | 1.24 (Pauling Scale) |
Chemical Properties of Erbium
- The metallic properties of erbium are characterized by its ability to maintain its lustrous appearance in dry atmospheric conditions while exhibiting a tendency to undergo tarnishing when exposed to moist air.
- The combustion of erbium metal is characterized by its propensity to readily react with oxygen, resulting in the formation of erbium(III) oxide.
- The chemical element erbium exhibits a reaction with water, resulting in the formation of its corresponding hydroxide compound. The reaction exhibits a sluggish response when exposed to cold water, but demonstrates an accelerated rate of reaction when subjected to hot water.
- The erbium metal exhibits reactivity with various halogens, resulting in the formation of trihalides that correspond to each respective halogen.
Chemical Reaction of Erbium
- The Reaction of Erbium With Air
Er exhibits a gradual oxidation process when exposed to atmospheric conditions, resulting in the formation of erbium(III) oxide, denoted as Er2O3, through a readily combustible reaction.
4 Er (s) + 3 O2 (g) → 2 Er2O3 (s)
- The Reaction of Erbium With Water
Er exhibits a sluggish reaction rate when exposed to cold water, while it undergoes a more rapid reaction when in contact with hot water. This reaction results in the formation of erbium hydroxide, denoted as Er(OH)3, along with the liberation of hydrogen gas (H2).
2 Er (s) + 6 H2O (g) → 2 Er(OH)3 (aq) + 3 H2 (g)
- The Reaction of Erbium With Halogens
The element erbium exhibits a propensity to engage in chemical reactions with various halogens, resulting in the formation of erbium (III) halides.
The chemical reaction between erbium metal and fluorine gas (F2) results in the formation of erbium (III) fluoride, denoted as ErF3.
2 Er (s) + 3 F2 (g) → 2 ErF3 (s) [Pink]
The chemical reaction between erbium metal and chlorine gas (Cl2) results in the formation of erbium (III) chloride, denoted as ErCl3.
2 Er (s) + 3 Cl2 (g) → 2 ErCl3 (s) [Violet]
The chemical reaction between erbium metal and bromine (Br2) results in the formation of erbium (III) bromide, denoted as ErBr3.
2 Er (s) + 3 Br2 (g) → 2 ErBr3 (s) [Violet]
The chemical reaction between erbium metal and iodine represented as I2, results in the formation of erbium (III) iodide, denoted as ErI3.
2 Er (s) + 3 I2 (g) → 2 ErI3 (s) [Violet]
- The Reaction of Erbium With Acid
Erbium exhibits high solubility in dilute sulphuric acid, leading to the formation of Er(III) ions and the liberation of hydrogen gas, H2. The solution exhibits a yellow coloration and is highly probable to correspond to the complex [Er(H2O)9]3+.
2 Er (s) + 3 H2SO4 (aq) → 2 Er3+ (aq) + 3 SO42- (aq) + 3 H2 (g)
Uses of Erbium
Numerous significant applications exist for erbium and its compounds some of which are discussed here:
Used In Alloys
It is occasionally combined with vanadium. An alloy is formed through the melting and combining of two or more metallic elements. The combination exhibits distinct characteristics that diverge from those exhibited by the individual metallic elements. Alloys composed of vanadium and erbium exhibit a relatively low level of hardness yet demonstrate a notable degree of workability. At cryogenic temperatures, erbium-nickel alloys exhibit an exceptional specific heat capacity.
Used In Lasers
Erbium-doped yttrium aluminum garnet (Er: YAG) lasers find extensive utility in diverse medical applications due to their pronounced affinity for water absorption. Laser surgeries necessitate the shallow deposition of the laser within the tissues. Erbium is utilized in medical lasers based on yttrium aluminum garnet (YAG) for various dermatological procedures, including skin resurfacing for conditions such as acne scars, mole removal, tattoo removal, and wart removal.
Used In Optics
- Erbium-doped fiber amplifiers (EDFAs) are extensively employed in the field of optical communications. These amplifiers are composed of an active element consisting of erbium-doped optical silica glass.
- An optical fiber refers to a slender, filamentous structure composed of glass or plastic materials that facilitates the efficient transmission of light. Light is responsible for transmitting messages through fiber optic cables, similar to how electricity transmits messages through copper telephone wires.
- Erbium optical fibers are utilized in long-distance communication systems and military applications for the transmission of messages. Telephone service providers are currently undergoing a transition from copper phone lines to optical fibers in order to enhance the quality and clarity of communication.
- Optical fibers possess a significantly higher capacity for transmitting information in comparison to traditional copper bundles.
Used in Nuclear Reactors
Erbium finds applications in nuclear reactors as control rods for neutron absorption or as a burnable poison within the design of nuclear fuel. In recent studies, erbium has been employed in experimental investigations pertaining to lattice confinement fusion. Erbium is employed in nuclear reactors for its role as a control rod that effectively absorbs neutrons.
The Health Effects of Erbium
- Erbium does not possess a recognized biological function, despite observations suggesting its potential to stimulate metabolic processes.
- Quantifying the quantity of erbium within the human body poses a challenging task. The concentrations of these substances are most prominent in skeletal structures, while relatively lesser quantities are found in the hepatic and renal organs.
Environmental Effects of Erbium
- Erbium does not present any discernible environmental hazards to the flora and fauna.
References
- https://www.rsc.org/periodic-table/element/68/erbium
- https://www.britannica.com/science/erbium
- https://www.chemicool.com/elements/erbium.html
- https://byjus.com/chemistry/erbium/
- https://www.thoughtco.com/erbium-facts-er-element-606531
- https://pubchem.ncbi.nlm.nih.gov/element/Erbium

