The term “wastewater” refers to any body of water whose quality has been diminished as a result of human activity. Wastewater is made up of both liquid and solid waste that is discharged from residential and commercial properties, industrial operations, and agricultural facilities or land. Wastewater can be classified as either hazardous or non-hazardous. Levels of a wide variety of contaminants can be found in wastewater in a variety of quantities.
Interesting Science Videos
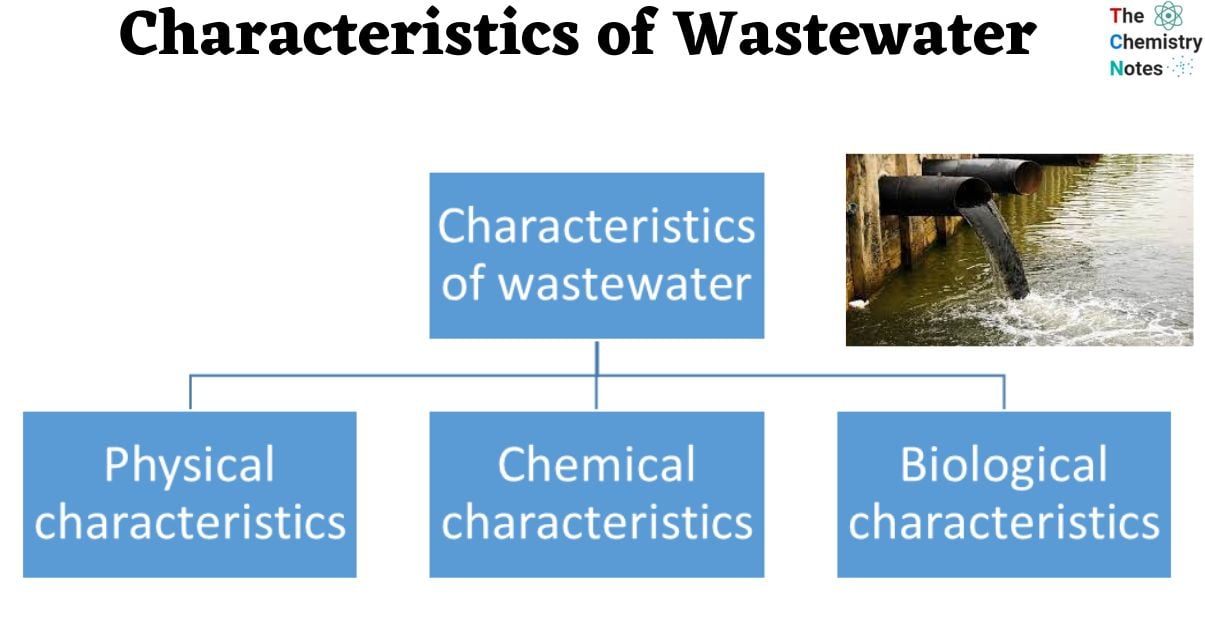
Characteristics of Wastewater
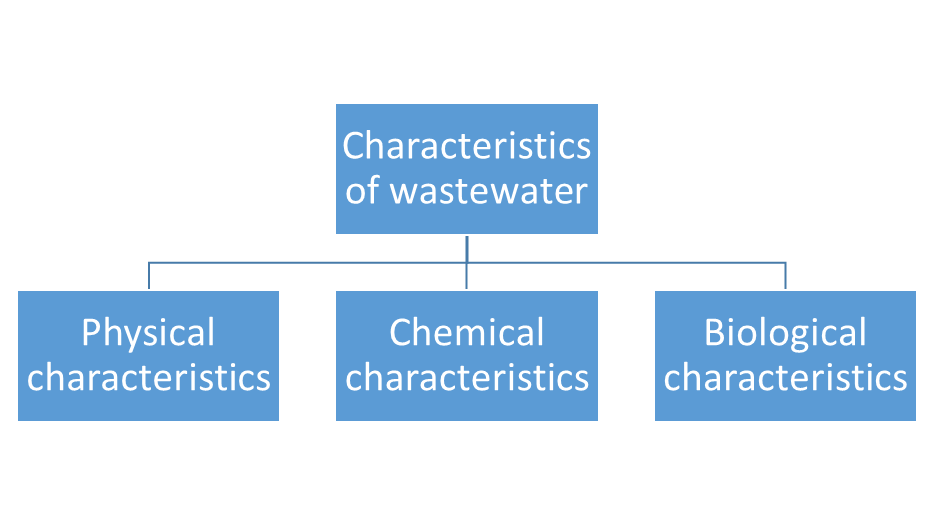
Physical Characteristics of Waste Water
Color: Fresh domestic sewage exhibits a grey coloration, bearing a resemblance to a diluted solution of soap. As time elapses, the process of putrefaction initiates, resulting in a darkening effect. Septic sewage typically exhibits a predominantly black or dark hue. The hue of industrial wastewater is contingent upon the specific chemical processes employed within industrial settings. The combination of industrial wastewater and domestic sewage has the potential to introduce pigmentation to the latter.
Odor: Fresh sewage typically possesses a musty odor that is generally not deemed offensive. However, as the sewage ages and becomes stagnant, it gradually emits an offensive odor. Once all the available oxygen in the sewage is depleted, it begins to release a malodorous combination of hydrogen sulfide gas and other sulfur compounds, which are generated by anaerobic microorganisms.
Temperature: Wastewater temperatures are often higher than municipal water temperatures because of the addition of warm water from homes and factories. The temperature of the wastewater increases much more as it circulates in closed circuits. Because of this, water becomes thicker and more conducive to bacterial growth. The typical temperature of Indian wastewater is near the optimal temperature for biological activities, at roughly 20 degrees Celsius.
Turbidity: The turbidity of wastewater is dependent upon the quantity of suspended solid particles within the system. Turbidity serves as an indicator of the light-emitting characteristics of wastewater, and the turbidity test is employed to assess the quality of waste discharges in relation to colloidal matter. The level of turbidity is contingent upon the concentration of sewage or wastewater. The turbidity of sewage increases proportionally with its strength or concentration. The measurement of turbidity can be conducted using either a turbidity rod or Jackson’s turbidimeter.
Total solids: A typical sample of sewage will have a water content of 99.9 percent and a solids content of 0.1 percent. The total solids content (ST) of wastewater is defined analytically as all the stuff that is left behind as residue after evaporation at temperatures between 103 and 105 degrees Celsius. There are three distinct manifestations of total solids that can be found in wastewater: (a) suspended particles; (b) colloidal solids; and (c) dissolved solids.
Chemical Characteristics of Wastewater
pH value: The acidity or alkalinity of wastewater can be determined by measuring its pH value. Raw sewage has a pH between 7.3 and 7.5, making it somewhat alkaline. However, with time, bacterial acid production causes the pH value to drop, and the sewage to become more acidic. A somewhat stable oxidation process, however, causes it to return to its original alkaline state.
Effluent that has been properly oxidized should have a pH of around 7.3. High levels of acids (pH 7) or alkalis (pH 7) in wastewater are characteristic of chemical wastes. There are treatment processes that rely on wastewater having a specific pH value, therefore knowing what that value is is crucial. Lime is sometimes used to achieve an alkaline state.
Chloride content: The presence of chloride in natural water is the result of the leaching of chloride-containing rocks and soils into the water from the surrounding environment. The chlorides that are found in home sewage come from various sources, including human feces, culinary waste, and urine discharges.
Nitrogen content: Nitrogen in sewage is a clear sign of decaying organic materials. Nitrogen, often known as a nutrient or bio-stimulant, is crucial for the development of Protista and plants. Protein synthesis relies on nitrogen, so measuring nitrogen levels is necessary for determining whether or not waste water can be treated biologically.
- Ammonia Nitrogen or Free Ammonia: The initial phase of organic matter decomposition is observed. In an aqueous solution, it can be found in either the form of an ammonium ion or ammonia, depending on the pH level.
- Organic Nitrogen: The Kjeldahl method is what is used to determine it. The total amount of ammonia and organic nitrogen is referred to as the Kjeldahl nitrogen.
- Albuminoid Nitrogen: The amount of nitrogen that is already present in wastewater before the process of breaking down organic matter may begin. It gives an indication of the quantity of nitrogenous material in the wastewater that is not fully constituted.
- Nitrites Nitrogen: Nitrites are an indicator that there is organic debris that has partially degraded.
- Nitrates nitrogen: Nitrates are an indicator of the existence of organic matter that has been entirely oxidized.
Fats, grease, and oils: The majority of it comes from waste products in the kitchen, such as butter, vegetable oils, and fats. In addition to this, waste from commercial establishments such as garages, workshops, and factories is included. They create problems with maintenance and interfere with the action of biological systems.
Surfactants: Synthetic detergents are the primary source of surfactants. These are the byproducts of the use of indoor plumbing, appliances, and electronics. Large organic molecules called surfactants (or surface-active agents) produce foaming during wastewater treatment. This prevents wastewater from being naturally aerated. In particular, the nonbiodegradability of the surfactant alkyl-benzene-sulphonate (ABS), which is widely employed in synthetic detergents.
Since its usage in detergents has been outlawed in some nations, biodegradable linear-alkyl-sulphonate (LAS) has mostly replaced ABS. A standard solution of methylene blue dye is used to quantify the amount of surfactants present in wastewater.
Phenols, pesticides & agricultural chemicals: Wastewater from factories is typically rich in phenols. Such wastewaters produce major taste problems in drinking water, especially when chlorination is used to disinfect the water. However, at concentrations up to 500 mg/l, phenols can be physiologically oxidized.
Surface runoff from farms, forests, and parklands is a major contributor to the presence of pesticides, herbicides, and other agricultural chemicals in a combined sewerage system. The carbon-chloroform extract method is used to determine the concentration of these microorganisms.
Toxic compounds: Copper, lead, silver, chromium, arsenic, and boron are some of the cations that are known to be toxic to microorganisms, which can contribute to the failure of biological treatment facilities. Boron is another cation that is known to be harmful. These are the byproducts of wastewater from industrial processes. The presence of some harmful anions in certain industrial wastes, such as cyanides and chromates, also makes it difficult for wastewater treatment facilities to do their jobs. Because of this, their presence needs to be taken into account while designing biological treatment plants.
Sulfates, Sulphides, and H2S Gas: Sulphates and sulfides are generated as a result of the decomposition process of diverse sulfur-containing substances found within wastewater. Sulfate ions (SO4) are naturally present in the majority of water supplies, and as a result, they are also found in wastewater. Sulfur, an essential element in protein synthesis, is liberated during the process of degradation.
The production of hydrogen sulfide gas results in a smelly and offensive byproduct. In addition to this, H2S gas undergoes biological oxidation to produce sulphuric acid, which results in the corrosion of sewer pipes. When sulfates are converted to sulfides, the biological process that takes place in sludge digesters is significantly hampered, particularly when the concentration of the sulfides is more than 200 mg/l.
Other gases: (i) Nitrogen gas (N2), (ii) Oxygen gas (O2), (iii) Carbon dioxide gas (CO2), (iv) Hydrogen sulfide gas (H2S), (v) Ammonia, chemically represented as NH3, and methane, represented as CH4, are two distinct compounds. The initial three gases present in the atmosphere are universally found in all bodies of water, including wastewater, that are exposed to air. Conversely, the latter three gases are produced as a consequence of the breakdown of organic substances that are present within the wastewater.
The anaerobic decomposition of organic matter in wastewater results in the production of methane gas, which serves as the primary by-product. The gas under consideration exhibits a lack of color and scent, while also possessing a high propensity for combustion. Due to the elevated risk of explosion, it is imperative to ensure adequate ventilation in manholes, sewer junctions, junction chambers, and similar infrastructural components.
Oxygen consumed: Consumed oxygen is the oxygen used in the breakdown of organic carbon. Adding a standard amount of potassium permanganate with diluted sulphuric acid to a wastewater sample yields the oxygen content. At a constant temperature of 18°C, we let the reaction proceed for 15 minutes and 4 hours.
The oxygen released by the potassium permanganate is used by the wastewater. This test is performed in place of the BOD test to establish the strength of the sewage (i.e., whether it is very strong, somewhat strong, or very weak). However, the total oxygen required for the biological oxidation of all or most of the organic materials is not provided by this assay.
Dissolved oxygen: The amount of oxygen that is in a dissolved condition in the wastewater is referred to as the dissolved oxygen, or DO. In most cases, wastewater does not have DO; nonetheless, the presence of DO in wastewater that has not been treated shows that the effluent is still fresh. In a similar vein, the presence of this compound in treated wastewater or effluent is an indication that a significant amount of oxidation has been achieved throughout the treatment stages.
It is crucial to verify that there is at least 4 ppm of DO present in the treated wastewater before discharging it into receiving waterways. It’s conceivable that fish and other aquatic species will perish in the disposal area if dissolved oxygen levels are low. Having DO in wastewater is good since it stops bad smells from forming.
Oxygen demand: The existence of oxygen is necessary for the continued functioning of living beings. After a certain point, aerobic action stops since there is no longer any oxygen in the wastewater; after that point, putrefaction starts to occur as a result of anaerobic action. Because of this, there is a requirement for oxygen in wastewater in order to facilitate the oxidation of both inorganic and organic materials.
Biological Characteristics of Wastewater
Sewage from homes typically includes quite large numbers of different kinds of microorganisms due to the nature of the material. There is a correlation between the existence of these microorganisms and the biological features of sewage. There is a correlation between the existence of microorganisms and the biological characteristics of wastewater.
- Aquatic plants
- Aquatic animals
- Aquatic bacteria and viruses
Anaerobic processes
- The process by which organic matter is broken down is known as putrefaction, and the end product, which occurs when enzymes cause solid organic matter to dissolve, is known as liquefaction.
- In the process of oxidizing organic materials, anaerobic bacteria utilize electron acceptors in addition to oxygen. In the course of carrying out their metabolic process, they produce carbon dioxide, water, hydrogen sulfide, methylamine, nitrogen, reduced organics, and additional bacteria.
- Septic tanks, Imhoff tanks, and sludge digestion tanks are examples of treatment units that rely only on putrefaction as their primary mechanism.
Aerobic processes
- Oxidation is the process by which aerobic bacteria complete their activity, which involves the combination of substances with oxygen.
- Electrons can only be accepted by aerobic bacteria if they have access to oxygen in the free state. The byproducts of aerobic action are carbon dioxide, water, sulfurous oxide, nitrogenous oxide, ammonia, and more microorganisms.
- Despite the fact that both of the processes described above work in the opposite direction—the first by breaking apart, and the second by constructing—there is coordination between the two.
- In the first stage of the process, the anaerobic bacteria break down the complex organic matter into simple organic compounds. Then, in the second stage, the aerobic bacteria oxidize those chemicals to generate stable compounds.
Treatment Processes of Wastewater
The composition of urban wastewater is predominantly organic in nature. Numerous industries, such as pharmaceuticals, chemical, and food processing sectors, exhibit substantial organic waste burdens, necessitating treatment procedures primarily focused on organic removal. In a conventional treatment facility, the wastewater undergoes a sequential treatment process using physical, chemical, and biological mechanisms, each designed to achieve targeted reductions in waste load. The aforementioned steps encompass a series of processes.
Pre-treatment of Waste Water
- Pre-treatment refers to the series of processes that constitute the established approach for the treatment of wastewater. This technology facilitates the elimination and fragmentation of large solid particles, the elimination of fine particles, and the segregation of stormwater. At this step of the treatment process, the removal of oil and grease is also undertaken.
Primary Treatment of Waste Water
- The initial stage of treatment is commonly referred to as sedimentation, clarifying, or settling system. The unit process under consideration involves the intentional retention of wastewater in a settling tank or reservoir for a designated duration. The wastewater undergoes a process wherein it is directed into a sedimentation tank that has been specifically designed for this purpose. The velocity at which the wastewater is transported through the tank is carefully controlled to allow for the settling of fine solids, which occurs due to the force of gravity acting upon them. Some of the benefits associated with it are:
Secondary Treatment of Waste Water
Following primary treatment, the clarified wastewater is combined with or exposed to a high concentration of microorganisms in an aerobic reactor.
- Suspended growth systems: It is described as those aerobic processes that obtain a high microbe concentration through the recycling of biological solids. This is what is meant by the term “biological solids recycling.”
- Attached growth systems: It makes it possible for a microbial layer to grow on the surface of the medium while at the same time allowing it to remain exposed to the atmosphere, the source of its oxygen supply. The wastewater is sprayed onto the layer of microorganisms that are present.
- Dual process systems: In order to accomplish its primary goal of obtaining very high-quality effluent requirements, it makes use of two-stage setups of fixed film and suspended growth processes.
Tertiary Treatment of Waste Water
- Prolonged settlement in the lagoons, irrigation of the grassland, or percolation regions (for treating the land) are all viable options.
- Passing the substance through a very tiny mesh or filtering it through sand or gravel.
References
- https://www.environmentalpollution.in/wastewater-management/characteristics-of-wastewater-wastewater-management/6476
- https://www.1h2o3.com/en/learn/wastewater-parameters/what-are-the-characteristics-of-wastewater
- https://www.schoolofpe.com/blog/2018/03/basic-characteristics-of-wastewater.html#:~:text=Odor%20%2D%20Wastewater%20that%20includes%20sewage,a%20higher%20turbidity%2C%20or%20cloudiness.
- https://allaboutcivil.org/characteristics-of-wastewater-physical-chemical-biological/
- https://www.onlinebiologynotes.com/physical-chemical-and-biological-characteristics-of-sewage/
- https://www.biologydiscussion.com/waste-management/waste-water-treatment/waste-water-characteristics-and-its-treatment/25131
