Atomic Mass Definition
Atomic mass is the mass of an atom or an isotope which includes the total mass of the nucleus and the electrons present in the periphery.
- Most of the contribution is made by the nucleus as it contains the heavier subatomic particles, while lesser contributions are made by the electrons and nuclear binding energy.
- The SI unit of atomic mass is the kilogram (kg), but it is often expressed in terms of non-SI unit, Dalton. 1 Dalton is defined as the 1/12th of the mass of a single carbon-12 atom.
- The atomic mass of a chemical species is often slightly less than the sum of the masses of protons, neutrons, and electrons due to the loss of some energy due to binding energy mass loss.
- Atomic mass represents the mass of an atom which can only be one isotope. This allows precise measurement of different atoms in contrast to the abundance-weighted average, like in relative atomic weight.
- Atomic mass can also be used for the determination of molecular mass, which differs slightly in numerical value from the molar mass.
Read Also: Atom vs Molecule- Definition, 12 Major Differences, Examples
Atomic Weight Definition
Atomic weight, also called relative atomic mass, is the weighted arithmetic mean of the relative isotopic mass of all the isotopes of a particular element depending on the abundance of each of those isotopes.
- Besides, atomic weight can also be related to the sum of the total number of protons and neutrons present in an atom.
- The value of atomic weight might be slightly different than the atomic mass as a substance contains different isotopes that have different atomic or isotopic mass.
- Atomic weights are expressed in terms of atomic mass units (AMU), which are also called daltons.
- Atomic weight is fundamental to chemistry as the most chemical reaction takes place via numerical relationships between atoms.
- Since the determination of the number of atoms involved in a reaction is quite difficult, most calculations are made by measuring the reactants and products to obtain information through atomic weight.
- Atomic weight is more common as it is more convenient and results in consistency throughout different regions of the world.
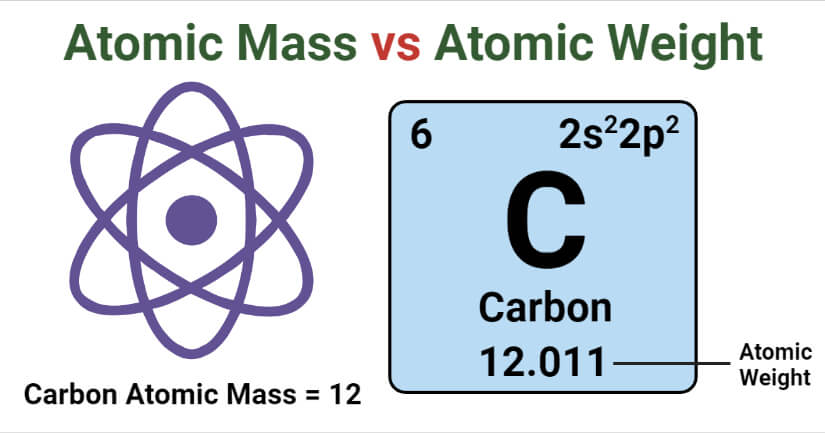
7 Key Differences (Atomic Mass vs Atomic Weight)
| Characteristics | Atomic mass | Atomic weight |
| Definition | Atomic mass is the mass of an atom or an isotope which includes the total mass of the nucleus and the electrons present in the periphery. | Atomic weight, also called relative atomic mass, is the weighted arithmetic mean of the relative isotopic mass of all the isotopes of a particular element depending on the abundance of each of those isotopes. |
| Calculation | Atomic mass is calculated by measuring the masses of protons, neutrons, and electrons of the atom. | Atomic weight is calculated by determining the percentage abundance of all the isotopes of a chemical element. |
| Isotopes | Atomic mass doesn’t depend on the isotopic mass of different isotopes of an element. | Atomic weight depends on the isotopic mass of different isotopes of an element. |
| Prevalent | Atomic mass is less prevalent than atomic weight. | Atomic weight is more prevalent in chemistry due to its use in the calculation of different chemical quantities involved in reactions. |
| Molecules | The sum of the atomic masses of all the atoms present in a molecule gives the molecular mass. | The sum of the atomic weights of all the atoms present in a molecule gives the molecular weight. |
| SI unit | The SI unit of atomic mass is the kilogram, but the non-SI unit, AMU, is often used. | The SI unit of atomic weight is AMU or daltons. |
| Value | The value of atomic mass is usually a whole number. | The value of atomic weight may or may not be a whole number. |
Atomic Mass vs. Atomic Weight (Video By MedSchoolCoach)

References and Sources
- 4% – https://www.calculatoratoz.com/en/atomic-mass-calculator/node-342?FormulaId=309
- 3% – https://www.britannica.com/science/atomic-weight
- 2% – https://www.thoughtco.com/atomic-weight-and-atomic-mass-difference-4046144
- 2% – https://en.wikipedia.org/wiki/Atomic_mass
- 1% – https://www.thoughtco.com/how-to-calculate-atomic-mass-603823
- 1% – https://www.onlinemathlearning.com/relative-atomic-mass.html
- 1% – https://www.minichemistry.com/atomic-mass-molecular-mass.html
- 1% – https://www.differencebetween.com/difference-between-atomic-mass-unit-and-vs-atomic-mass/
- 1% – https://quizlet.com/7200704/chemistry-4-flash-cards/
- 1% – https://physics.stackexchange.com/questions/332972/why-was-carbon-12-chosen-for-the-atomic-mass-unit
- 1% – https://pages.mtu.edu/~pcharles/SCIHISTORY/Atomic_weight_.html
- 1% – https://en.wikipedia.org/wiki/Standard_atomic_weight
- 1% – http://dictionary.sensagent.com/Molecular%20mass/en-en/
- <1% – https://www.thoughtco.com/experimental-determination-of-avogadros-number-602107
