Astatine is a chemical element with an atomic number of 85 and is represented by the symbol ‘At’ in the periodic table. Its appearance is unknown classified as metal and belongs to the p-block of group 17 of the periodic table. It is the rarest naturally occurring element in the Earth’s crust, only appearing as a byproduct of the breakdown of different heavier elements.
Astatine, along with boron(B), arsenic (As), antimony (Sb), silicon (Si), germanium (Ge), and tellurium (Te), is occasionally included in the list of metalloids, albeit its characteristics have not been well researched due to its short half-life.
History of Astatine
- Walter Minder examined the radioactivity of a certain radium sample and claimed that it included a new element; subsequently, he performed chemical experiments on the sample and discovered that the new element is chemically comparable to iodine.
- Horia Hulubei and Yvette Cauchois felt they had found an unfamiliar element in 1939 by using a high-resolution X-ray apparatus to examine several mineral samples, but they decided against doing further research.
- The final known halogen, astatine, was synthesized in 1940 at the University of California by Corson and peers. The word astatos, which means “unstable” in Greek, describes this radioactive substance.
- Dale R. Corson, Kenneth Ross MacKenzie, and Emilio Segrè were able to produce astatine for the first time in 1940 by bombarding bismuth with alpha particles.
Occurrence of Astatine
- Astatine is synthesized by blasting bismuth with alpha particles. It can be produced naturally by thorium or uranium decay.
- The Earth’s crust contains around 2.36 x 1025 grams of astatine, which is less than 1 gram.
- There are now 39 known isotopes of astatine, and theoretical modeling shows that there may be 37 additional isotopes.
- All of the astatine isotopes discovered so far have very short half-lives, with 210At having the longest at 8 hours 10 minutes.
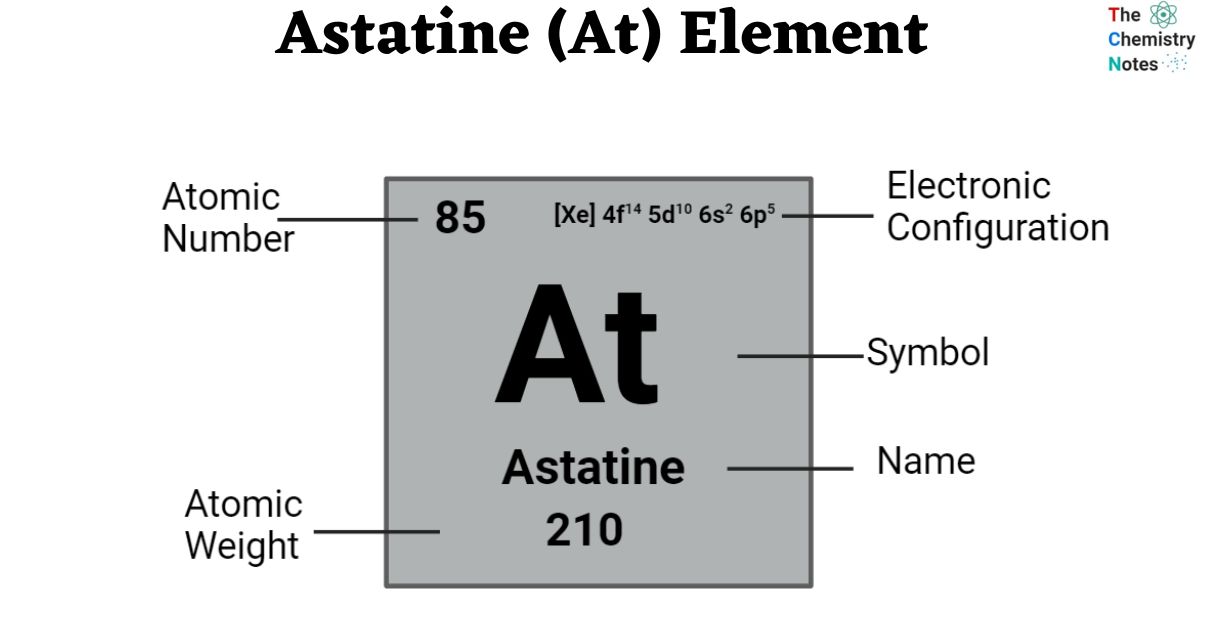
Elemental Properties of Astatine
| Electronic Configuration | [Xe] 4f14 5d10 6s2 6p5 |
| Atomic Number | 85 |
| Atomic Weight | (210), no stable isotope |
| State at 20°C | Solid |
| Group, Period, and Block | 17, 6, p-block |
| Density | 7 g/cm3 at 20 °C (approx.) |
| Appearance | Unknown |
| Van der Waals radius | unknown |
| Electron shells | 2, 8, 18, 32, 18, 7 |
| Electrons | 85 |
| Protons | 85 |
| Neutrons in the most abundant isotope | 125 |
Physical Properties of Astatine
- The majority of astatine’s physical characteristics have been calculated theoretically.
- Astatine has an atomic number of 85 and is a grey metal. It has a melting point of 302 °C and a boiling point of 377 °C.
- The accurate density of astatine is unknown but it is estimated to be 7 g/cm3 at 20 °C (approx.).
- The structure of astatine is unknown; it is anticipated to have the same structure as iodine: orthorhombic crystalline.
- Astatine is really amazing. When exposed to room temperature, half of the astatine vaporizes after one hour.
- There are now 39 known isotopes of astatine, and theoretical modeling shows that there might be 37 additional isotopes.
Chemical Properties of Astatine
- The chemical properties of astatine are only studied by use of tracer testing.
- Astatine is the least chemically reactive of the halogens and has the most metallic characteristics of the halogen group.
- Astatine, like the other halogens, is predicted to form salts with metals such as sodium.
- Astatine may also combine with hydrogen to generate hydrogen astatide, which when dissolved in water forms hydro-astatic acid.
- Astatine compounds with an oxidation state of “+1” have characteristics of silver compounds with the same oxidation state.
- It has been shown that astaxanthin and EDTA can create complexes.
- Astatine’s organic chemistry characteristics are similar to those of iodine.
Uses of Astatine
There is no industrial usage for astatine. Some other uses are mentioned here:
- 211At is occasionally used as a radioactive tracer and in cancer therapy.
- Astatine is released in the human thyroid gland because it functions similarly to iodine. As a result, it is utilized in the treatment of thyroid illnesses.
Health Hazard of Astatine
- The entire quantity of astatine in the earth’s crust at any given moment is less than 30 grams, and just a few micrograms have ever been intentionally manufactured.
- This, along with its short lifespan, eliminates the need to examine the implications of astatine on human health.
- Astatine is examined in a few nuclear research facilities, where its extreme radioactivity necessitates unique handling procedures and safeguards.
Environmental Effects of Astatine
- Astatine does not exist in considerable quantities in the biosphere and hence does not generally pose a concern.
Video Reference
References
- https://chemicalengineeringworld.com/astatine-element-properties-and-information/
- https://www.rsc.org/periodic-table/element/85/astatine
- https://www.lenntech.com/periodic/elements/at.htm
- https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Supplemental_Modules_and_Websites_(Inorganic_Chemistry)/Descriptive_Chemistry/Elements_Organized_by_Block/2_p-Block_Elements/Group_17%3A_The_Halogens/Z085_Chemistry_of_Astatine_(Z85)
- https://www.chemistrylearner.com/astatine.html

