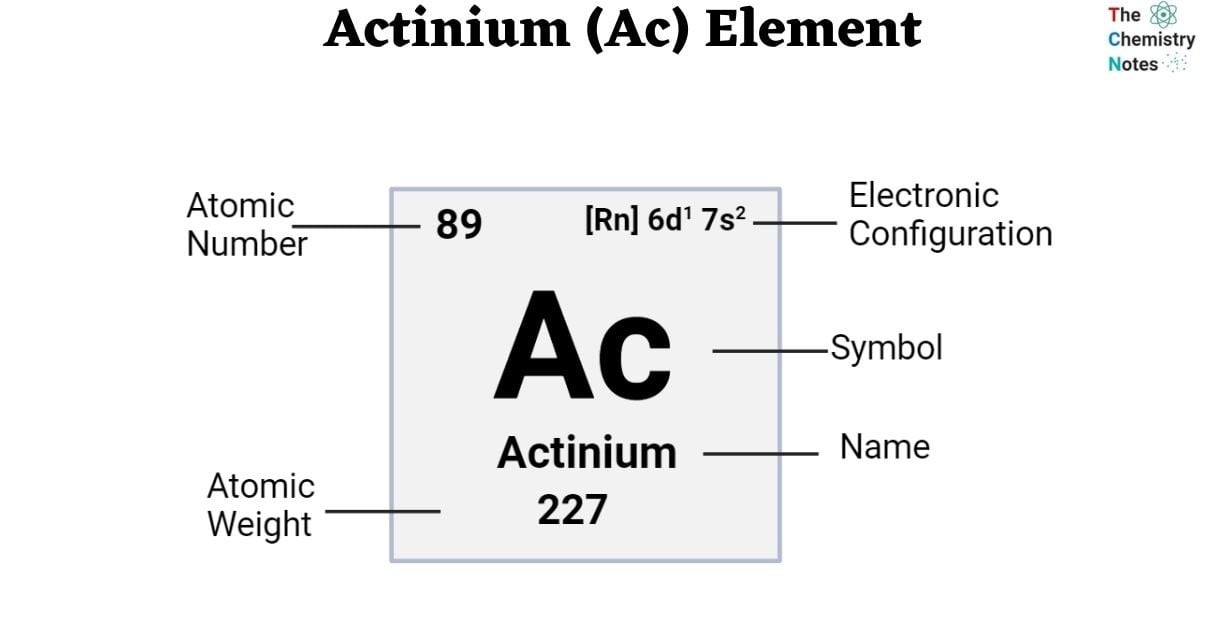
The atomic number of actinium is 89. It is found in f-block and Period 7 of the periodic chart. The symbol ‘Ac’ represents it. The name is derived from the ancient Greek word ‘aktis’, meaning beam or ray. Actinium is found only in trace amounts in uranium and thorium ores. It is soft, silvery-white in appearance. Actinium, like most lanthanides and many actinides, has an oxidation state of +3 in virtually all of its chemical compounds. Ac is one of 15 elements in the periodic table that form the actinoid series.
Interesting Science Videos
History of Actinium
- Debierne and Marie Curie’s joint research led to the discovery of actinium in the uranium mineral pitchblende, from which radium and polonium had already been recovered.
- He was able to detect the existence of a new element since the radioactive emission profile of his sample could not be explained by the presence of any known element.
- Debierne was unable to separate pure actinium from the thorium that was also present in pitchblende.
- In 1899, while researching novel ways to separate rare earth oxides in Paris, Debierne recovered the element from uranium-rich pitchblende ores.
- Friedrich Otto Geisel, who was not aware of Debierne’s discovery, independently extracted the element and called it emanium in 1902.
- Upon comparisons, Debierne’s name, Actinium, was kept due to its seniority. The term actinium comes from the Greek words “aktis” or “aktinos,” which means “ray” or “beam.”
Occurrence of Actinium
- The element is found in uranium ore deposits generated by the radioactive decay of uranium and its other radioisotopes. Its level of concentration in the earth’s crust is estimated to be 0.0005 parts per trillion by mass.
- Actinium metal has been synthesized by reducing Ac fluoride with lithium vapors at temperatures within the range of 2012 °F to 2372 °F.
- Actinium is usually created by neutron irradiation of the radium isotope 226Ra within a nuclear reactor due to its scarcity in nature.
- Actinium has 36 radioactive isotopes, with half-lives that range from 69 nS for the shortest (217Ac) to 21.77 years for the longest (227Ac).
Elemental Properties of Actinium
| Electronic Configuration | [Rn] 6d1 7s2 |
| Atomic Number | 89 |
| Atomic Weight | (227) g.mol -1, no stable isotope |
| State at 20°C | Solid |
| Group, Period, and Block | 7, f-block |
| Density | 10.07 g/cm3 at 20 °C |
| Ionic radius | unknown |
| Van der Waals radius | unknown |
| Electron shells | 2, 8, 18, 32, 18, 9, 2 |
| Electrons | 89 |
| Protons | 89 |
| Neutrons in most abundant isotope | 138 |
Physical Properties of Actinium
- Actinium is a metal with qualities comparable to lanthanum, the element just above it on the periodic table.
- Ac has an atomic number of 89. It has a melting point of 1227 °C (2240 °F) (estimated) and a boiling point of 3200±300 °C (5800±500 °F) (extrapolated).
- Ac has a solid phase density of 10.07 g/cm3 near room temperature.
- It is a delicate, silvery-white, radioactive metallic element that glows in the dark with a faint blue light.
- Its calculated shear modulus is comparable to lead.

Chemical Properties of Actinium
- When exposed to air, the surface of actinium reacts instantly with either oxygen (O) or moisture in the air to generate a thin crust of white actinium oxide, preventing further oxidation.
- It weighs around 150 times more than radium.
- Actinium’s most frequent oxidation state is +3. Actinium compounds exhibit characteristics comparable to lanthanum compounds.
- It is highly electropositive and is likely to generate a variety of allotropes.
- Because of its radioactivity, actinium chemistry is challenging to investigate.
- Actinium forms oxides, halides, oxyhalides, and oxo phosphates.
Chemical Reaction of Actinium
- The Reaction of Actinium with Air
Actinium metal readily transforms into actinium (III) oxide, Ac2O3, in moist air.
4 Ac (s) + 3 O2 (g) → 2 Ac2O3 (s)- The Reaction of Actinium with Carbonate
Carbonate ions precipitate Ac3+.
2 Ac3+ (aq) + 3 CO32− (aq) → Ac2(CO3)3 (s)- The Reaction of Actinium with Oxalate
Oxalate ions precipitate Ac3+.
2 Ac3+ (aq) + 3 C2O42− (aq) → Ac2(C2O4)3 (s)- The Reaction of Actinium with Phosphate
Phosphate ions precipitate Ac3+.
Ac3+ (aq) + PO43− (aq) → AcPO4 (s)Uses of Actinium
Despite being an excellent generator of alpha rays, it has no particular commercial uses. Here are a few examples of different uses:
- Actinium is used to treat cancer in two ways: as a reusable generator and as a direct agent in radiation treatment.
- Actinium is utilized as a component of the neutron probe, which detects the amount of water in the soil and also analyzes moisture for quality control during highway building.
- Because actinium-227 has a half-life of 21.77 years, it may be used to simulate the vertical mixing of slow oceanic water. A velocity analysis does not give the same level of precision as a concentration-depth profile.
Health Hazards of Actinium
- Because actinium is highly radioactive, it should be handled with caution and only by specialists and qualified personnel.
- Actinium-227 is exceedingly radioactive, and its potential for radiation-induced health consequences is comparable to that of plutonium. Even tiny doses of actinium-227 would pose a major health risk.
- Radioactivity harms the gene pool and impacts future generations.
- Even low-dose exposures are carcinogenic with prolonged exposure.
- If swallowed, it is deposited in the bones and liver, where radioactive decay harms cells, perhaps leading to bone cancer or other disorders.
Environmental Hazards of Actinium
- Actinium discharges in the environment, whether small or large, represent a hazard of accumulation and amplification in the food chain.
- Radiation traverses species and accumulates in the food chain, exposing additional animals and people to its harmful effects.
- The food chain connects plants, animals, and people. As a result, improper disposal endangers all life.
Video Reference
References
- https://www.rsc.org/periodic-table/element/89/actinium
- https://www.lenntech.com/periodic/elements/ac.htm
- https://www.chemicool.com/elements/actinium.html
- https://www.americanelements.com/actinium.html
- https://chemicalengineeringworld.com/actinium-element-properties-and-information/
- https://www.industrialheating.com/blogs/14-industrial-heating-experts-speak-blog/post/95360-facts-about-the-elements-actinium
- https://www.thoughtco.com/interesting-facts-about-actinium-603672
- https://www.chemistrylearner.com/actinium.html
- https://pilgaardelements.com/Actinium/Reactions.htm

