Yttrium is a metallic element with the atomic number 39 and is represented by the symbol ‘Y’ in the periodic table. It is classified as a transition metal and belongs to the d-block of group 3 of the periodic table. It is a soft metal with a silvery metallic luster and is relatively stable in the air.
Yttrium does not occur as a free element on the Earth’s crust. Its presence in the crust in rare earth mineral ores is 0.003 percent, which makes it the 28th most abundant element on Earth.
History of Yttrium
- Carl Axel Arrhenius, a part-time chemist from Sweden, discovered a huge black rock in a discarded pit in the Swedish settlement of Ytterby in 1787. He named it ytterbite and sent samples to Johan Gadolin for research, thinking it was a previously unidentified mineral with the recently identified element tungsten.
- Johan Gadolin, a Finnish chemist at the University of Abo, discovered the novel oxide “earth” in 1789; it was named earth because it included yttrium oxide (Y2O3), which could not be broken down further by burning with charcoal, and published his comprehensive study in 1794.
- Anders Gustaf Ekeberg, a Swedish analytical chemist, validated Gadolin’s discovery in 1797 and dubbed the new oxide Yttria.
- It was in the year 1828 that yttrium was first isolated as a metal by Friedrich Wöhler, the German chemist. He treated a volatile chloride, which he believed was yttrium oxide, with potassium.
- Carl Gustaf Mosander, a Swedish chemist, conducted a more detailed investigation of yttrium oxide in 1843 and learned that it consisted of three oxides: yttrium oxide (white), terbium oxide (yellow), and erbium oxide (rose-colored).
- Yttrium is named after the Ytterby (where the yttrium was first discovered)in Sweden.
- ‘Yt’ was used as the symbol for Yttrium up until the 1920s, which was later changed to “Y”.
Occurrence of Yttrium
- Yttrium can be found in practically every rare earth mineral, including uranium; however, it does not occur naturally as a free element.
- In the Earth’s crust, yttrium is a moderately abundant element. Its abundance ranges from 28 to 70 parts per million. That puts yttrium on par with cobalt, copper, and zinc in terms of abundance.
- The majority of yttrium is now derived through ion exchange from monazite sand. Monazite (Ce, La, Nd, Th) (PO4, SiO4) is a rare earth element-rich mineral.
- Other minerals that contain yttrium metal include bastnasite (La, Ce, Y)CO3F, fergusonite (YNbO4), and samarskite (Y Fe3+Fe2+U, Th, Ca)2(Nb, Ta)2O8.
- It is difficult to distinguish this element from other rare-earth elements. It is commercially made by reducing yttrium fluoride with calcium metal, but different methods are possible. When removed, it turns into a dark gray powder.
- Countries with the biggest yttrium reserves are China, India, Brazil, Australia, and The United States, among others. However, only a few tonnes of metal is produced currently.
Isotopes of Yttrium
Yttrium has only one naturally occurring stable isotopes; 89Y
Naturally occurring isotopes
| Isotopes | Natural abundance (atom %) |
|---|---|
| 89Y | 100 |
Elemental Properties of Yttrium
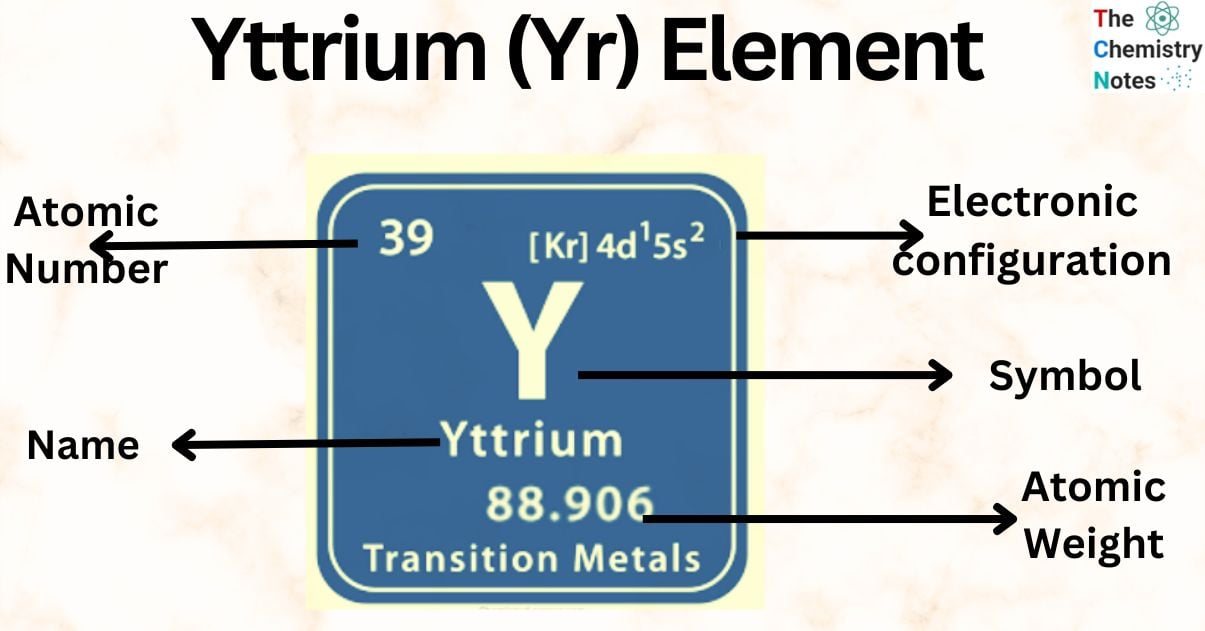
| Electronic Configuration | [Kr] 4d15s2 |
| Atomic Number | 39 |
| Atomic Weight | 88.906 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | 3, 5, d-block |
| Density | 4.47 g cm -3 at 20 °C |
| Covalent radius | 190(7) pm |
| Van der Waals radius | 0.106 nm (+3) |
| Electron shells | 2,8,18,9,2 |
| Electrons | 39 |
| Protons | 39 |
| Neutrons in most abundant isotope | 50 |
Physical Properties of Yttrium
- Yttrium has an atomic number of 39 and is silvery-white shiny metal. It has a melting point of 1522°C (2772°F) and a boiling point of 3345°C (6053°F).
- The density of yttrium is 4.47 grams per cubic centimeter.
- It is recognized as a paramagnetic element because of its unusual crystalline structure and internal electronic configuration; it becomes magnetic once subjected to an external magnetic field.
- It is an intermediate conductor (whose conductivity is in between those of metal and an insulator) of electricity and heat.
- Yttrium is malleable, allowing it to be easily hit into sheets without cleavage, and ductility, which makes it possible to draw thin wires from it.
- Yttrium is odorless.
| Color/physical appearance | Silvery white |
| Melting point/freezing point | 1522°C, 2772°F, 1795 K |
| Boiling point | 3345°C, 6053°F, 3618 K |
| Density | 4.47 g cm-3 at 20° |
| Electronegativity | 1.22 (Pauling Scale) 1.12 (Allen Scale) |
Chemical Properties of Yttrium
- Yttrium’s chemical characteristics are similar to those of rare earth elements.
- It reacts with cold water slowly and with hot water very rapidly.
- Yttrium reacts with acids and alkali.
Chemical Reaction of Yttrium
- The reaction of Yttrium with Air
In the air, yttrium metal tarnishes slowly and quickly burns to generate yttrium (III) oxide, Y2O3.
4Y (s) + 3O2 (g) → 2Y2O3 (s)
- The reaction of Yttrium with Water
When finely split or heated, yttrium dissolves in water, producing solutions containing the aquated Y(III) ion and hydrogen gas, H2.
2Y (s) + 6H2O (aq) → 2Y 3+ (aq) + 6OH– (aq) + 3H2 (g)
- The reaction of Yttrium with Halogens
Yttrium is highly reactive towards the halogens.
Yttrium reacts with fluorine, F2, and burns to form the trihalide yttrium (III) fluoride, YF3.
2Y (s) + 3F2 (g) → 2YF3 (s)
Yttrium reacts with chlorine, Cl2, and burns to form the trihalide chloride, YCl3.
2Y (s) + 3Cl2 (g) → 2YCl3 (s)
Yttrium reacts with bromine, Br2, and burns to form the trihalide yttrium (III) bromide, YBr3.
2Y (s) + 3Br2 (g) → 2YBr3 (s)
Yttrium reacts with iodine, I2, and burns to form the trihalide yttrium (III) iodide, YI3.
2Y (s) + 3I2 (g) → 2YI3 (s)
- The reaction of Yttrium with Acid
Yttrium metal rapidly dissolves in dilute hydrochloric acid to generate solutions containing the aquated Y(III) ion and hydrogen gas, H2.
2Y (s) + 6HCl (aq) → 2Y3+ (aq) + 6Cl– (aq) + 3H2 (g)
Uses of Yttrium
Yttrium is used in different sectors some of which include:
Used In Ceramics and Speciality Glasses: Yttrium oxide is utilized in several ceramic and glass formulas because of its high melting point, shock resistance, and low thermal expansion. It is also used in optical glasses and camera lenses due to its extraordinary characteristics.
Used In Electronics:
Television
The most common application of yttrium is in the production of red phosphorus, which produces white and gray colors in television cathode ray tubes and LEDs.
When europium (Eu) is combined with yttrium oxide (Y203), yttrium orthovanadate (YVO4), or yttrium oxysulfide, red phosphorus is produced.
LED’s
It is also utilized in tri-chromatic fluorescent lighting to provide magnificent white light while saving significant energy.
Microwave Filter
Yttrium is used to make yttrium-iron garnets, which are used to filter microwaves for radar to block certain frequencies and are widely used in communication devices and satellites.
Used As Alloy: It’s used to make magnesium and aluminum alloys stronger. To lower the grain size of chromium, molybdenum, zirconium, and titanium alloys, small amounts of yttrium are added. The addition of yttrium to alloys improves their workability and increases their resistance to high-temperature recrystallization. It is utilized as a metal on the electrodes of some high-performance spark plug.
Some Other Uses Of Yttrium
Yttrium barium copper oxide is extremely important because it is utilized in the manufacturing of high-temperature superconductors that conduct electricity with no energy loss.
Small amounts of yttrium are utilized in the cathodes of lithium ion phosphate batteries to increase capacity and durability.
Health Effects Of Yttrium
- Because moisture and fumes can be inhaled, yttrium is especially dangerous in the workplace.
- Yttrium can also cause cancer in humans if inhaled, as it raises the risk of lung cancer.
- When it accumulates in the body of an individual, it might prove harmful to the liver.
Environmental Effects of Yttrium
Yttrium can enter the environment when domestic equipment is discarded. Yttrium will progressively accumulate in soils and water, eventually leading to increased concentrations in humans, animals, and soil particles.
In aquatic animals, yttrium induces cell membrane damage, which has a number of detrimental impacts on reproductive as well as nervous system functions.
References
- Per Enghag, Encyclopedia of the elements: technical data, history, processing, applications., John Wiley and Sons, 2004, p433,434.
- https://www.chemicool.com/elements/yttrium.html
- https://www.rsc.org/periodic-table/element/39/yttrium
- https://pubchem.ncbi.nlm.nih.gov/element/Yttrium#section=Ionization-Energy
- https://www.lenntech.com/periodic/elements/y.htm
- W. M. Haynes, ed., CRC Handbook of Chemistry and Physics, CRC Press/Taylor and Francis, Boca Raton, FL, 95th Edition, Internet Version 2015, accessed December 2014.
- Tables of Physical & Chemical Constants, Kaye & Laby Online, 16th edition, 1995. Version 1.0 (2005), accessed December 2014.
- J. S. Coursey, D. J. Schwab, J. J. Tsai, and R. A. Dragoset, Atomic Weights and Isotopic Compositions (version 4.1), 2015, National Institute of Standards and Technology, Gaithersburg, MD, accessed November 2016.
- T. L. Cottrell, The Strengths of Chemical Bonds, Butterworth, London, 1954
- https://www.osti.gov/biblio/5122889

