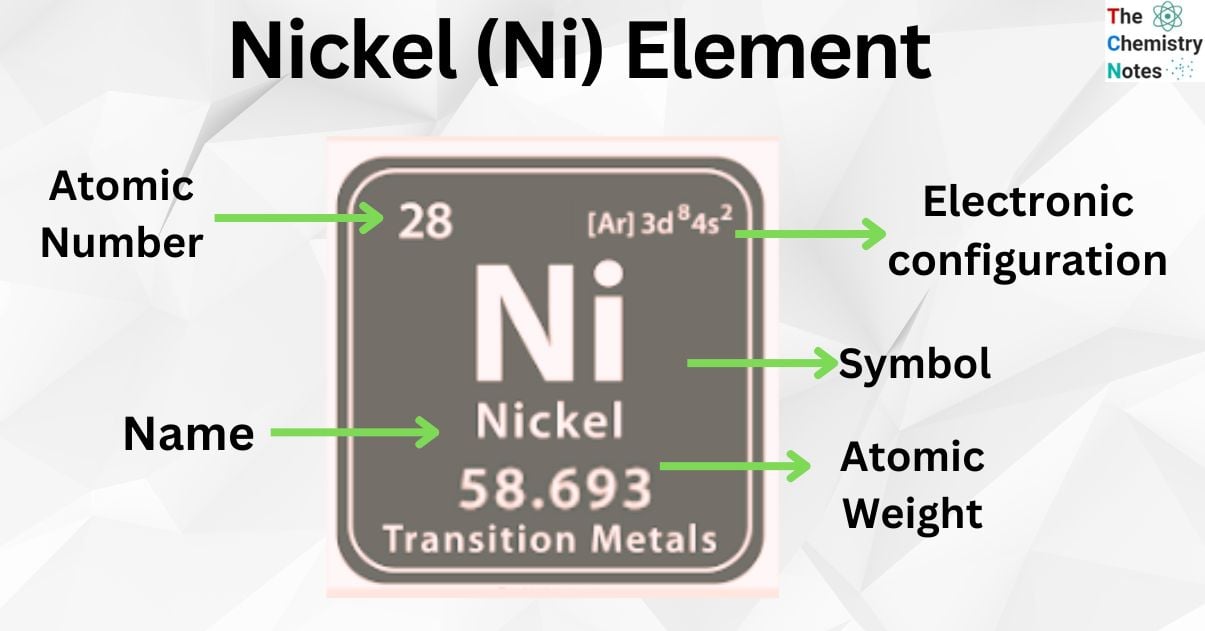
Nickel is the metallic element with the atomic number 28 and is represented by the symbol ‘Ni’ in the periodic table. It is classified as a transition metal and belongs to the d-block of group 10 (X) of the periodic table. It is a silvery-white lustrous metal with a slight golden tinge which has important magnetic properties.
Nickel is thought to be the second most common element in the earth’s core, right behind iron. It is 22nd most abundant element in Earth’s crust. Overall it is the 5th most abundant element on Earth. Magnetic sulfide deposits and laterite deposits, which are the by products of severe weathering of nickel-rich rocks on the surface, are the two main types of deposits where nickel typically occurs.
History of Nickel
- Nickel has been used throughout history by ancient civilizations all across the world, whether or not they were aware of it.
- Nickel has been accidentally used for centuries, dating all the way back to 3500 BCE. As much as two percent nickel was discovered in bronzes from what is currently known as Syria.
- Axel Fredrik Cronstedt, a scientist at Stockholm, discovered an unknown element in 1751 that came from a mine near Los, Hälsingland, Sweden and is now known as nickeline (NiAs).
- He believed it may include copper, but what he ultimately identified as nickel by 1754 turned out to be brand-new metal.
- Its elemental nature was established in 1775 when Torbern Bergman a Swedish chemist was able to create pure nickel.
- The scarce Kupfernickel first served as the only source of nickel. Nickel was produced as a byproduct of cobalt blue manufacture starting in 1824.
- In the year 1881 Switzerland became the first country to originally introduce coins made of almost pure nickel .
- Nickel gets its name from the German word used in miner’s mythology ‘Kupfer-nickel’ meaning devil’s copper or St. Nicholas’s copper.
- French scientist Joseph-Louis Proust, who was based in Spain at the time and performed his research there, first discovered nickel in meteorites in 1799.
Occurrence of Nickel
- Natural nickel comprises roughly 0.008% of the Earth’s crust. The majority of nickel is believed to be found in the cores of Earth and other terrestrial planets, where it reacts with iron and other light elements to create alloys, according to geophysical and geochemical evidence. The majority of current models place its mass proportion in the Earth’s core at about 5.2%.
- Two different types of ore deposits are extracted for the majority of nickel. The first is laterite, where the main ore mineral mixes are (Fe, Ni)O(OH), garnierite (a mixture of several hydrous nickel and nickel-rich silicates), and nickeliferous limonite.
- The second is magmatic sulfide deposits, where pentlandite (Ni, Fe)9S8 is the key ore.
- Massive nickel deposits have been suggested to exist in the deep water. Nickel-abundant manganese nodules are found on the deep-sea floor.
- The latest estimate of the world’s nickel reserves is close to 350 million tons.
- More than half of the world’s nickel deposits are found in Australia, Indonesia, South Africa, Russia, and Canada. Nickel is concentrated economically in laterite- and sulfide-type ore deposits.
Isotopes of Nickel
There are five stable isotopes of Nickel (Ni) that are present in nature: 58Ni, 60Ni, 61Ni, 62Ni, and 64Ni.
Naturally occurring isotopes
| Isotope | Natural abundance (atom %) |
|---|---|
| 58Ni | 68.0769 (89) |
| 60Ni | 26.2231 (77) |
| 61Ni | 1.1399 (6) |
| 62Ni | 3.6345 (17) |
| 64Ni | 0.9256 (9) |
- Cu-64, which is utilized in radioimmunotherapy, is created from Ni-64.
- Most stable Nickel isotopes have been used to study human absorption of Nickel.
Elemental Properties of Nickel
| Electronic Configuration | [Ar] 3d8 4s2 |
| Atomic Number | 28 |
| Atomic Weight | 58.693 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | 10, 4, d-block |
| Density | 8.90 g.cm -3 at 20 °C |
| Ionic radius | 0.069 nm (+2) ; 0.06 nm (+3) |
| Van der Waals radius | 0.124 nm |
| Electron shells | 2, 8, 16, 2 |
| Electrons | 28 |
| Protons | 28 |
| Neutrons in most abundant isotope | 30 |
Physical Properties of Nickel
- Nickel has an atomic number of 28 and is a hard silvery metal. It has a melting point of 1455 °C (2651 °F) and a boiling point of 2913 °C (5275 °F).
- The density of Nickel is 8.9 grams per cubic centimeter.
- It is recognized as a ferromagnetic element. ‘Ni’ is one of only three metal elements that are naturally magnetic (the others being iron, Fe, and cobalt, Co).
- Nickel is ductile (it can be pulled out into thin wires) and malleable to some extent (it can be pressed into thin sheets).
- Nickel serves as an excellent electrical conductor. Because electrons in nickel are free to move around they are able to carry electrical charge from one end to other.
- Nickel is an excellent thermal conductor as well. Heat causes a metal’s particles to vibrate more rapidly and move around more swiftly. Energy is transferred from one particle to another as they come into contact.
- Nickel is odorless and tasteless.
| Color/physical appearance | silver-white |
| Melting point/freezing point | 1455°C, 2651°F, 1728 K |
| Boiling point | 2913°C, 5275°F, 3186 K |
| Density | 8.90 g.cm-3 at 20°C |
| Malleability | Yes |
| Ductility | Yes |
Chemical Properties of Nickel
- Nickel doesn’t combine with oxygen or water or dissolve in most acids at room temperature because it is a relatively less reactive metal. It becomes more reactive at higher temperatures.
- The metal can be highly polished and does not tarnish in air.
- In terrestrial and meteoric deposits, elemental nickel and iron are only extremely infrequently found combined.
Chemical Reaction of Nickel
- Reaction of Nickel with Water
Nickel metal does not react with water under normal conditions.
- Reaction of Nickel with Air
Under normal conditions, nickel doesn’t react with air. However, when it is finely split metal reacts well with air and can become pyrophoric during the period.
The reaction of metal with oxygen (O2) seems to slow down at higher temperatures and create some nickel (II) oxide instead.
2Ni(s) + O2(g) → 2NiO(s)
- Reaction of Nickel with the Halogens
Fluorine gas, F2, and nickel metal do interact, however, the reaction is sluggish. As a result, nickel is a valuable metal for fluorine storage tanks.
Nickel metal reacts with chlorine (Cl2), bromine(Br2), or iodine(I2) to produce dichloride, NiCl2, dibromide, NiBr2, and diiodide, NiI2.
Ni(s) + Cl2(g) → NiCl2(s) [yellow]
Ni(s) + Br2(g) → NiBr2(s) [yellow]
Ni(s) + I2(g) → NiI2(s) [black]
- Reaction of Nickel with Acids
When nickel metal is slowly dissolved in diluted sulfuric acid, solutions containing the ionized Ni(II) and hydrogen gas, H2, are created. In practice, the complex ion [Ni(OH2)6]2+ represents the Ni(II).
Ni(s) + H2SO4(aq) → Ni2+(aq) + SO42-(aq) + H2(g)
Uses Of Nickel
One of the most sought-after and used metals in contemporary industry is Nickel. Nickel (Ni) is a transition metal that is used in numerous applications. What distinguishes it from other metals is its adaptability. There are many other uses for it, but only a handful are covered below.
Used As Superalloy
Nickel is being used as a superalloy, a type of high-temperature metal substance that can function under specific stress conditions. Corrosion-resistant high-temperature alloys with nickel bases are frequently employed at service temperatures over 500°C. Since their introduction in the 1950s, nickel-base superalloys have been widely employed for high-temperature applications in aerospace, power generation, and automotive.
Different types of nickel based alloys are :
Inconel Alloy : Nickel makes up the biggest portion of the elements that are utilized in Inconel alloys, with chromium coming in second. These alloys can withstand extreme stresses, are resistant to corrosion and oxidation, and can be used in harsh situations. It is used for the fabrication of combustion chambers, thermal reactors, insulating cans, refractory cans, gas turbines, chemical production, and many more.
Cupro Nickel : It has excellent thermal conductivity, is incredibly ductile, has exceptional tensile strength, and is particularly resistant to corrosion caused by seawater. It is used for military equipment ,piping and heat exchangers ,desalination plants ,propellers and propeller shafts,etc.
Brushed Nickel : Despite being corrosion-resistant, brushed nickel tarnishes quickly It may get a milky white tarn as it ages and is exposed to the elements. It is used to make kitchen faucets, bathroom fittings, cabinet hardware,lighting fixtures etc.
Invar Alloy : This alloy of iron and nickel has a small coefficient of thermal expansion as its main characteristic. It is mostly utilized in clocks, seismic creep gauges, valves in engines, large aerostructure molds, etc.
Nichrome Alloy : This nickel-chromium alloy has excellent electrical resistance as well as elevated temperature resistance as its main characteristics. Its color is silvery-gray. Its primary applications include resistance wire, heating elements for appliances like space heaters and toasters, and dental restorations.
There are other many more nickel based alloys that are used in our daily lives.
Uses Of Nickel in Electronics : Electronic equipment and electric cars that employ nickel batteries frequently contain nickel. This metal is also used in large electronic components and the development of nanotechnology. Parts of laptops and smartphones contain nickel. Nickel-coated layers can also be found on compact discs.
Uses Of Nickel in Aerospace, Marine and Military : The First and second world wars, as well as the subsequent cold wars, brought nickel to attention. Weapon manufacturing increased throughout this time, and different countries engaged in fierce warfare to establish their control.The use of nickel-based alloys is on the rise in the aircraft sector as well. Warships, aircraft armor plates, and tank armor all require a particular use of nickel. NASA has used it to protect spacecraft from the sun’s heat. It has a great resistance to corrosion, making it perfect for use in the construction of marine vehicles.
Uses Of Nickel In Cars
Nickel is most commonly used in cars, the stainless steel found in cars is nickel alloy. due to the high corrosion resistance auto industry depends on stainless steel. It also has the ability to absorb energy from crashes. It is also lighter and more sturdy when compared to other metals.
Use Of Nickel In Electroplating
Nickel is used in electroplating. It increases wear resistance and inhibits corrosion. The overall thickness of the plate is additionally increased. Nickel improves the appearance and brightness of an object from an aesthetic standpoint. It is also used as a base layer for gold and silver.
Health Effects Of Nickel
Nickel is a hard, silver-white metal that can be found in soil, water, and foods like almonds, dried beans, and chocolate. Nickel is used by humans for a wide variety of purposes. A very small amount of nickel is required by the body. In several bodily chemical reactions, nickel is a necessary nutrient.Its particular bodily roles are not well understood.
- Nickel is more readily absorbed through the lungs of smokers. Nickel can enter the human body through the air we breathe, the water we drink, the food we eat, and cigarette smoke. Nickel is necessary in modest amounts, but if it is consumed in excessive amounts, it can be harmful to human health.
- Nickel fumes can irritate the lungs and lead to pneumonitis. Individuals who are sensitive to nickel and its compounds may develop the dermatitis known as “nickel itch” as a result of exposure.
- The following effects result from excessive absorption of nickel:
- Increased likelihood of developing cancer of the prostate, laryngeal cancer, nose cancer, and malignancies of the lungs
- Feeling unwell and lightheaded after inhaling nickel gas
- Lung embolism
- A respiratory condition
- Defective births
Environmental Effects of Nickel
Power plants and waste incinerators emit nickel into the natural environment. All nickel compounds will, to some degree, adsorb to sediment or soil particles and, therefore, remain sedentary once they are discharged into the environment. However, nickel is bound to get more permeable in the acidic ground and frequently rinse out into the groundwater.
Information on nickel’s effects on species other than humans is not very abundant. We are aware that elevated nickel concentrations in sandy soil can obviously hurt plants while elevated nickel concentrations in surface waters can inhibit the growth of algae.Microorganisms can also experience growth reduction when nickel is present, but they often become resistant to nickel over time.
What is nickel? What is so special about this essential metal? Where does it come from? And what is it used for? With this video, we take you on a quick tour of the world of nickel.
References
- Mary Elvira Weeks, The discovery of the elements. III. Some eighteenth-century metals., J. Chem. Educ., 1932
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- https://www.lenntech.com/periodic/elements/ni.htm
- https://www.rsc.org/periodic-table/element/28/nickel
- https://pubchem.ncbi.nlm.nih.gov/compound/Nickel#section=Uses
- https://www.britannica.com/science/nickel-chemical-element#ref224861
- https://www.chemicool.com/elements/nickel.html
- J. D. Lee, Concise Inorganic Chemistry, 5th Edition, John Wiley and Sons. Inc. 2007.
- F. A. Cotton, G. Wilkinson & C. Gaus, Basic Inorganic Chemistry, 3 rd Edition, John Wiley & Sons (Asia), Pvt., Ltd., 2007.

