Carbon nanotubes (CNT) are the fundamental building blocks of nanotechnology. Nanotechnology heavily relies on carbon, which has an atomic number of six. While researching the graphite electrode surface used in electric arc discharge, Iijima unintentionally discovered them. This unintentional discovery laid the groundwork for the exciting field of nanotechnology and launched a new direction in carbon research. A carbon nanotube (CNT) is a hexagonal array of carbon atoms rolled up into a long, thin, hollow cylinder known for its size, shape, and extraordinary physical properties. They can be chemically and physically manipulated in material science, electronics, energy management, biomedical applications, and many other fields.
What are Carbon Nanotubes?
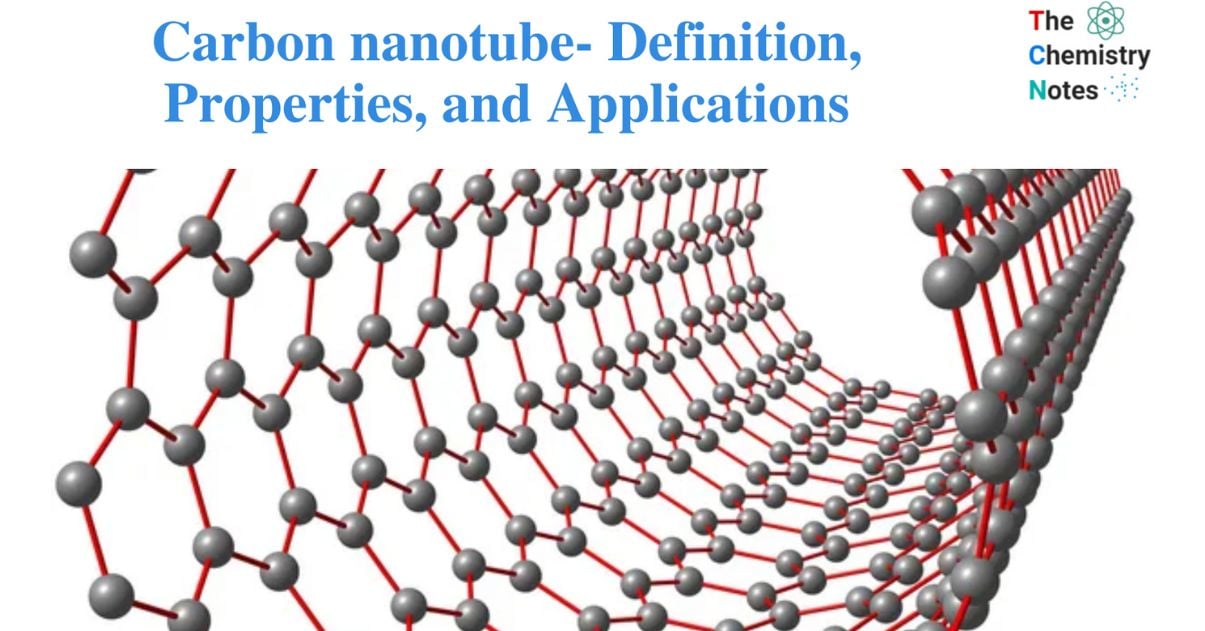
Carbon nanotubes (CNTs) are cylindrical molecules made of rolled-up sheets of single-layer carbon atoms (graphene).
They can be single-walled (SWCNT) with a diameter of less than 1 nanometer (nm) or multi-walled (MWCNT) with diameters greater than 100 nm. Their length can range from a few micrometers to several millimeters.
A carbon nanotube is a carbon allotrope that resembles a tube of carbon atoms.
Carbon nanotubes are one of the most studied nanomaterials due to their exceptional mechanical, electrical, and thermal properties. They are extremely strong and difficult to break, but they are still light.
Structure of Nanotube
A carbon nanotube is a tube-shaped material made of carbon with a nanometer-scale diameter. Carbon nanotubes are classified into two types: single-walled nanotubes (SWNT) and multi-walled nanotubes (MWNT).
Single-Walled Nanotubes (SWNT)
They have a diameter of about 1 nanometer and a tube length that can vary millions of times. They can be formed in three shapes based on the rolling of graphene sheets into a seamless cylinder: armchair, zigzag, and chiral. Each form has a distinct effect on the electrical properties of the nanotube.

Multi-walled Nanotubes (MWNT)
The discovery of C60 prompted the researchers to look for other carbon compounds that contained curved graphenes. This resulted in the discovery of multi-walled nanotubes constructed from concentric cylinders of rolled-up graphene sheets capped with semi-fullerenes. The tube length was a few meters, and the diameter was 10-20 nm. These structures exhibit properties similar to fullerenes and graphite as they grow in size.

Carbon Nanotube Properties
Tensile strength
Carbon nanotubes are stronger than steel and Kevlar. This strength is derived from sp2 bonds formed between individual carbon atoms. Carbon nanotubes are not only strong but also pliable. When a force is applied to a nanotube, it can bend and then return to its original shape when the force is removed. The elasticity of a nanotube has a limit, and under extremely strong forces, it is possible to deform the shape of a nanotube permanently. Flaws can reduce the strength of a nanotube in its structure. Atomic vacancies or a rearrangement of the carbon bonds cause defects. Defects in the structure can weaken a small segment of the nanotube, causing the tensile strength of the entire nanotube to weaken. The tensile strength of a nanotube, like the strength of a chain, is determined by the strength of the weakest segment in the tube.
Electrical Properties
Carbon nanotubes have a conducting nature due to the sp2 bonds formed between carbon atoms. Because of the strength of the bonds, they can also withstand high electric currents. When used as interconnects on semi-conducting devices, single-walled nanotubes can route electrical signals at speeds of up to 10 GHz. Their electronic properties can be manipulated by using an external magnetic field, mechanical force, etc.
Thermal Stability
Carbon nanotubes can withstand high temperatures, making them excellent thermal conductors. These nanotubes are considered temperature stable up to 28000°C and around 750°C in air. Carbon nanotubes have been shown to transmit more than 15 times the amount of watt per minute than copper wires.
Nanotube Synthesis
Carbon nanotubes can be prepared using various methods such as arc discharge (AD), laser ablation, chemical vapor deposition (CVD), and some of the more recent methods that use high-pressure carbon monoxide or a unique catalytic mixture. Carbon nanostructures such as fullerenes, graphene, and nanotubes are of particular interest in both current research and future industrial applications. Because the band gap of single-walled carbon nanotubes (SWNTs) can range from zero to about 2 eV, their electrical conductivity can be that of a metal or a semiconductor.
Applications of Carbon Nanotubes
Carbon nanotubes have numerous potential applications due to their remarkable properties. They can be used in electronics, textiles such as water and tear-resistant fabric, sensors based on thermal conductivity, and many other applications. They have exceptional heat and electrical conductivity properties, making them a good candidate for various applications. The following are some of the most important applications of carbon nanotubes.
Transistors
Transistors are the foundation for modern integrated circuits that function as digital switches. Different carbon-nanotube configurations produce defects that allow single-walled nanotubes to function as transistors. Nanotube-based switches the size of a single electron had been envisioned, but they required cryogenic temperatures.
A molecule can be placed inside a carbon nanotube to affect the electronic current flowing through it in such a switch. The end result is a molecular-scare gate in which the molecule’s position controls the flow of electrical current. The gate in this model is about one nanometer in size, which is three orders of magnitude smaller than a silicon chip.
Sensor
Carbon nanotubes have been reported to be a good gas sensor due to their elongated shape. They measured the DC electrical resistance and thermoelectric power of SWNT bundles and thin films.
Nanoelectronics
Because of their high conductivity, nanotubes have the potential to be used in the field of electronics. Single-walled nanotubes are the more conducting of the two types. Nanotubes become highly conducting when they are twisted and bent. Nanotubes, with their high conductivity and small size, could be an alternative to copper, which is commonly used but ineffective at sizes smaller than 40 nm.
Batteries
Rechargeable lithium-ion batteries are used in the majority of portable electronic devices. When lithium ions move between two electrodes, one of which is graphite and the other is metal oxide, the batteries release charge. Carbon nanotube electrodes can be ten times thinner and lighter than amorphous carbon electrodes, and their conductivity is over a thousand times greater. In some cases, such as electric vehicles, weight reduction can significantly reduce battery power requirements. Carbon nanotubes were used to create supercapacitors with a power density of 30kw/kg (compared to 4kw/kg for commercially available devices). Supercapacitors of this type could drastically reduce the time it takes to recharge devices like laptops and cell phones.
Barrier to Carbon nanotube success
There are several remaining technical and non-technical barriers to CNT success. Covering these in detail would be lengthy and likely impossible; on the other hand, such a passage would be a path to success with nanotubes! However, here are a few major roadblocks.
Heterogeneity in Electronics
The fact that batches of nanotubes are heterogeneous mixtures of metallic and semiconducting tube types is one issue with nanotube production for electronics. Electrical devices typically require these types to be separated, but tuning production in this regard has been difficult. Doping, or tuning conductivity, and electrical behavior at contact points are also issues.
Orientation
Controlling the orientation of nanotubes is a problem where recent progress has been made. Nanotubes are commonly grown in a disorganized manner (affectionately known as a “rat’s nest”), making them difficult to use in microprocessors.
Dimensions and Density
The size of manufactured nanotubes varies greatly. Nanotube manufacturers will need to make size more consistent for commercial use. Despite the fact that nanotubes are extremely thin, nanotube matrices typically have a relatively large (100nm) spacing between tubes.
References
- Carbon Nanotubes-Properties and Applications- Organic & Medicinal Chemistry International Journal DOI: 10.19080/OMCIJ.2018.07.555705
- https://www.nanowerk.com/nanotechnology/introduction/introduction_to_nanotechnology_22.php
- https://nanoscalereslett.springeropen.com/articles/10.1186/1556-276X-9-393
- https://www.vedantu.com/chemistry/carbon-nanotubes
- https://en.wikipedia.org/wiki/Carbon_nanotube
- https://byjus.com/chemistry /carbon-nanotubes/
- Kuzmych O, Allen BL, Star A (2007) Carbon nanotube sensors for exhaled breath components. Nanotechnology 18(37): 375502
