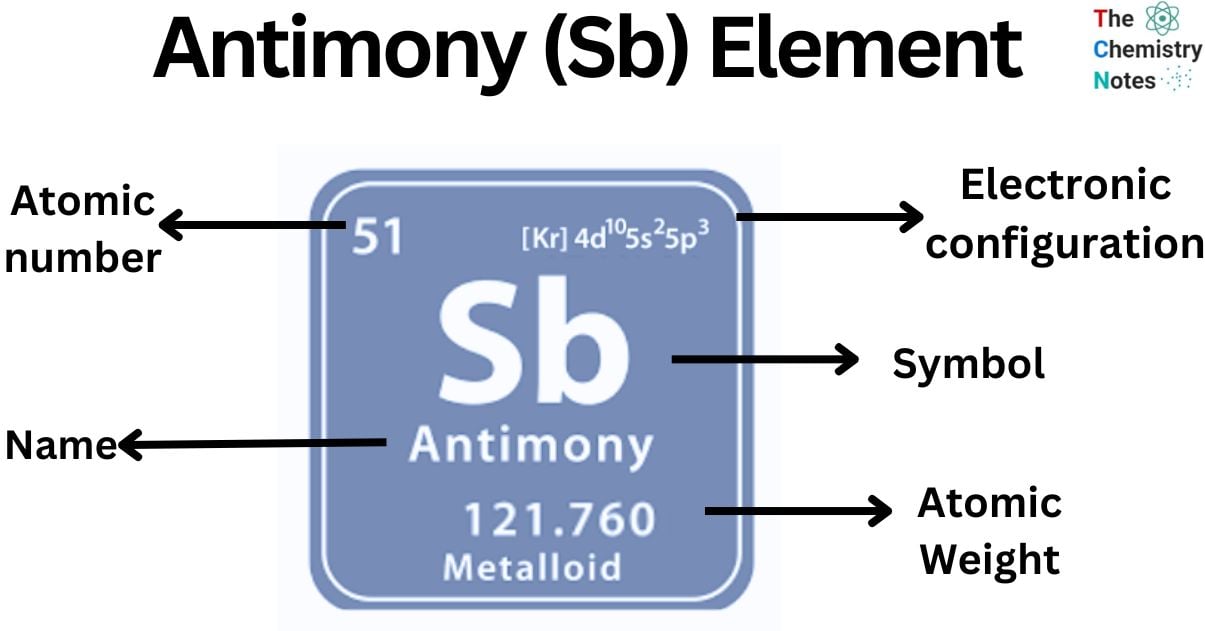
Antimony is a chemical element with the atomic number 51 and is represented by the symbol ‘Sb’ in the periodic table. It is classified as a metalloid and belongs to the p-block of group 15 of the periodic table. It is a lustrous silvery-white semimetal. It exists in different allotropic forms and is found in nature primarily as gray sulfide mineral stibnite (Sb2S3).
It is one of the rare elements its presence on Earth’s crust is estimated to be 0.2 ppm (parts per million). Despite its rarity, it is found in more than 100 different minerals.
Interesting Science Videos
History of Antimony
- Antimony has been used for thousands of years by humans. Artifacts dug from ancient civilizations date back more than 3000 years. They were familiar with both metallic and sulfide forms of antimony.
- Pieces of Chaldean vases dating back to 4000 BCE made of antimony have been dug by archaeologists that prove the metal’s use in ancient civilization.
- Stibnite (Sb2S3) was used as a ground powder for cosmetic purposes (especially eye makeup) by Egyptians around 3100 BCE.
- Another astonishing finding from Egypt dates back between 2500 BCE and 2200 BCE. It is a copper object plated with antimony.
- Around 77 AD, the Roman scholar Pliny the Elder (Gaius Plinius Secundus) detailed seven different pharmaceutical cures and many ways of preparing antimony sulfide for medical reasons in his work Natural History.
- The alloys used to make bells, and mirrors were known to have contained antimony as early as the 15th century.
- In 1615, Andreas Libavius, a German physician, documented the synthesis of metallic antimony via the direct reduction of the sulfide with iron.
- Nicolas Lémery’s 1675 release of a chemical textbook also provides information on the element’s synthesis processes.
- In 1783, a Swedish scientist and engineer named Anton von Swab announced the discovery of naturally occurring, pure antimony in the Earth’s crust.
Occurrence of Antimony
- Antimony is one of the rare elements its presence on Earth’s crust is estimated to be 0.2 ppm (parts per million). Despite its rarity, it is found in more than 100 different minerals.
- It exists in different allotropic forms and is found in nature primarily as gray sulfide mineral stibnite (Sb2S3).
- China, Russia, Myanmar, Tajikistan, and Australia are among the major producers of antimony.
Isotopes of Antimony
There are two naturally occurring stable isotopes of antimony: 121Sb and 123Sb.
Naturally Occurring Isotopes of Antimony
| Isotope | Natural abundance (atom %) |
|---|---|
| 121Sb | 57.21 (5) |
| 123Sb | 42.79 (5) |
Allotropes of Antimony
Antimony exhibits four allotropes, comprising of one metallic form and three metastable forms. The metallic allotrope exhibits stability, whereas the three metastable forms demonstrate instability. The metastable configurations manifest in the form of solid materials that exhibit yellow, black, and explosive properties.
Metallic Allotrope
The metallic allotrope of antimony is considered to be the most thermodynamically stable allotrope of this element. Antimony possesses a unique characteristic of exhibiting expansion upon freezing, a property shared by only a select group of elements, including bismuth. The metallic allotrope of antimony can be considered as the conventional manifestation of this element.
Metastable Forms
Metastable forms refer to the physical states of a substance that are not in their thermodynamically stable state, but can persist for a certain period of time due to kinetic barriers that prevent them from transitioning to a more stable state.
Explosive Allotrope
The explosive allotrope is a distinct form of a chemical element that exhibits explosive properties. The solid solution of antimony trichloride is known as the explosive allotrope of antimony. The object in question exhibits a high degree of sensitivity, such that even minor abrasions have the potential to initiate a catastrophic detonation. In 1858, George Gore, an electrochemist, successfully synthesized explosive antimony, marking the initial instance of its preparation.
Initially, a highly concentrated solution of antimony trichloride undergoes an electrolytic process within a hydrochloric acid solution. The electrolysis process necessitates the presence of both an anode and a cathode. Specifically, antimony serves as the anode, while either platinum or copper functions as the cathode. Subsequently, antimony is deposited onto the cathode, thereby posing a potential risk of explosion. In the event that antimony comes into contact with the surface of platinum/copper and causes abrasion, a transformation of antimony into a more stable metallic allotrope is likely to occur through the process of vaporization.
Upon vaporization of antimony trichloride, exothermic heat is liberated, resulting in the formation of a sudden burst of white clouds. The process in question poses a significant hazard due to the toxicity of antimony trichloride fumes. Upon inhalation, this substance elicits irritation in the oral cavity, nasal passages, pharynx, and pulmonary system. Furthermore, it has the potential to induce headaches and abdominal discomfort, while also posing a risk to the liver, cardiovascular system, and reproductive health.
Black Antimony
Antimony is a chemical element that is commonly known as black antimony due to its characteristic dark color. Black antimony is an amorphous substance that lacks a defined shape, and it is produced through the rapid cooling of antimony gas. Black antimony exhibits higher chemical reactivity in comparison to its metallic allotrope. Nevertheless, it exhibits greater instability and susceptibility to oxidation upon exposure to atmospheric oxygen, potentially leading to spontaneous combustion. Moreover, when exposed to temperatures of approximately 100˚C (212˚F), black antimony undergoes a transformation into its stable metallic state.
Yellow Antimony
The allotrope known as yellow antimony is classified as a nonmetal and is considered to be the least stable among its counterparts. The formation of the compound occurs through the oxidation of stibine (SbH3) at a low temperature of -90˚C (-130˚F). Upon exceeding a certain temperature threshold, yellow antimony undergoes a transformation into black antimony. Naples yellow, also referred to as yellow antimony, is an inorganic pigment commonly utilized in paint applications.
The color spectrum produced ranges from a deeper, reddish-yellow hue to a vivid, pale yellow hue. Naples yellow, which has been in use since the early 17th century, is considered to be one of the most ancient pigments utilized by individuals.
Elemental Properties of Antimony
| Electronic Configuration | [Kr] 4d10 5s2 5p3 |
| Atomic Number | 51 |
| Atomic Weight | 121.76 g.mol -1 |
| State at 20°C | Solid |
| Group, Period, and Block | 15, 5, p-block |
| Density | 6.697 g.cm -3 at 20 °C |
| Ionic radius | 0.245 nm (-3); 0.062 nm (+5); 0.076 nm (+3) |
| Van der Waals radius | 206 pm |
| Electron shells | 2, 8, 18, 18, 5 |
| Electrons | 51 |
| Protons | 51 |
| Neutrons in most abundant isotope | 71 |
Physical Properties of Antimony
- Antimony has an atomic number of 51 and is a silvery lustrous metalloid. It has a melting point of 630.63 °C (1167.13 °F) and a boiling point of 1635 °C (2975 °F).
- It has a solid phase density of 6.697 gm/cm3 and a liquid or molten phase density of 6.53 gm/cm3.
- Antimony is hard and brittle, meaning it can break easily.
- Antimony is a poor electrical conductor and thermal conductor.
- Antimony exists in four allotropic form : a stable metallic form, and three metastable forms (explosive, black, and yellow).
![Antimony [Image source: novaelements]](https://scienceinfo.com/wp-content/uploads/2023/06/image-129.png)
| Color/physical appearance | Lustrous, silver |
| Melting point/freezing point | 903.78 K (630.63 °C, 1167.13 °F) |
| Boiling point | 1908 K (1635 °C, 2975 °F) |
| Density | 6.697 g cm-3 at 20° |
| Malleability | No (It is brittle) |
| Ductility | No ( It is brittle) |
| Electronegativity | 2.05 (Pauling Scale) |
Chemical Properties of Antimony
- Antimony is typically unaffected by air and moisture, but in humid environments, it slowly transforms into an oxide. It can be oxidized by sulfur and halogens when heated.
- When heated in the air, antimony burns and emits a bright blue flame, which further gives off white fumes of the trioxide Sb2O3.
- Antimony trioxide is soluble in both acids and alkalis.
- Antimony has an electrical structure that is very similar to that of arsenic, with three outer shell orbitals that are only partially filled. As a result, it can display the +3 and -3 oxidation states and create three covalent bonds.
- Antimony has a lower electronegativity than arsenic. It can oxidize a variety of metals, forming antimonides that are similar to nitrides, phosphides, and arsenides but are a little more metallic in appearance.
Chemical Reaction of Antimony
Antimony’s stability makes it relatively inert at ambient temperatures. Air or water won’t cause it to react. But some halogens can trigger it to react. In addition, heated sulfuric and nitric acids are capable of dissolving antimony, but it is insoluble in water.
- The Reaction of Antimony With Air
Antimony interacts with oxygen in the air to generate the trioxide antimony(III) oxide, Sb2O3 when heated. The flame is bluish-white.
4 Sb (s) + 3 O2 (g) → 2 Sb2O3 (s)
- The Reaction of Antimony With Water
Antimony interacts with water at high temperatures to generate the trioxide antimony(III) oxide, Sb2O3 and hydrogen gas H2. At ambient temperature, antimony acts less rapidly.
2 Sb (s) + 3 H2O (g) → Sb2O3 (s) + 3 H2 (g)
- The Reaction of Antimony With Halogens
Antimony reacts with halogens under controlled conditions.
When antimony reacts with fluorine, F2, under controlled conditions, it forms the trihalide antimony (III) fluoride, SbF3.
2 Sb (s) + 3 F2 (g) → 2 SbF3 (s) [white]
When antimony reacts with bromine, Br2, under controlled conditions, it forms the trihalide antimony (III) bromide, SbBr3.
2 Sb (s) + 3 Br2 (g) → 2 SbBr3 (s) [white]
When antimony reacts with chlorine, Cl2, under controlled conditions, it forms the trihalide antimony (III) chloride, SbCl3.
2 Sb (s) + 3 Cl2 (g) → 2 SbCl3 (s) [white]
When antimony reacts with iodine, I2, under controlled conditions, it forms the trihalide antimony (III) iodide, SbI3.
2 Sb(s) + 3 I2 (g) → 2 SbI3 (s) [red]
- The Reaction of Antimony With Acids
Antimony dissolves in hot concentrated sulphuric acid, H2SO4, or nitric acid, HNO3, to form solutions containing Sb(III). The sulphuric acid reaction produces sulphur(IV) dioxide gas. Antimony does not react with hydrochloric acid in the absence of oxygen.
Uses Of Antimony
Since antimony’s physical characteristics are unsuitable for engineering, it is mostly employed as an additive in the metalworking industry. Here are some applications of antimony:
Used As Alloys
Its primary commercial application is as an alloying element for lead and some lead-based alloys to provide corrosion resistance and make the alloy durable and stiff. Tin-based babbitt and pewter, which are utilized in bearing metal applications, are made using antimony as an alloying component. Additionally, the element is commonly employed in the production of castings, soldiering supplies, and cable insulations. Batteries, type metals, low-friction metals, and other commercial goods are all produced using specific lead-antimony alloys.
Used In Flame Retardants
Antimony trioxide has a flame-retarding action because it forms halogenated antimony compounds, which interact with hydrogen atoms and possibly oxygen atoms and OH radicals to prevent fire. These flame-retardants can be found in children’s clothing, toys, and seat covers for cars and airplanes, among other products. Additionally, they are included in fiberglass composites made with polyester resins for components like light aircraft engine covers.
Used In Medicines
It offers few applications in biology and medicine. Antimonials, or medications containing antimony, are used as emetics. As antiprotozoal medications, antimony compounds are employed. Since 1919, potassium antimonyl tartrate often known as tartar emetic, was employed as an anti-schistosomal drug.
Some other uses of antimony includes
Some semiconductors, including infrared detectors and diodes, are made using pure antimony. Lead is hardened and made more durable using it. Antimony is used in batteries to ensure proper operation. Small firearms and ammunition are also made with it. It is also used in paints and ceramic enamels.
Health Effects of Antimony
- Those who work with the metal might experience the effects of exposure from breathing in antimony dust. By inhaling antimony-containing air, water, and food, as well as by coming into contact with antimony-containing soil, water, and other substances, humans can be exposed to the metal.
- The primary source of the health impacts is inhaling antimony, which is linked to hydrogen in the gaseous phase. Prolonged exposure to relatively high antimony concentrations (9 mg/m3 of air) can irritate the eyes, skin, and lungs.
- As the exposure continues, more serious health impacts may arise, such as lung diseases, heart issues, diarrhea, severe vomiting, and stomach ulcers. It is unclear whether antimony can induce cancer or reproductive failure.
- Antimony is used in medicine as a treatment for parasite infections, but people who consumed excessive amounts of the medication or were intolerant to it have encountered health consequences in the past. These health impacts have made humans more aware of the potential hazards of being exposed to antimony.
Due to the ubiquitous usage of antimony, employees must be completely protected by wearing overalls and respirators, regularly monitoring air quality, and regularly inspecting air extraction systems. Its biological surveillance during work is crucial.
Environmental Effects of Antimony
Antimony (Sb) is a poisonous metalloid, and as a result of its widespread use and associated Sb-mining activities, its pollution has emerged as a major environmental issue on a global scale. The chemical speciation of Sb has a significant impact on both its toxicity and mobility.
The release of Sb into the environment is caused by a combination of anthropogenic inputs and natural processes such weathering, biological activity, and volcanic activity. Sb released into the environment has the ability to adsorb on organic or inorganic environmental media, undergo redox reactions, change its form, and have harmful consequences on the ecosystem.
- Antimony can be found in soil, water, and air in extremely minute concentrations. Antimony will mostly pollute soils. Through groundwater, it can migrate enormous distances toward other sites and surface waterways.
- Studies in the laboratory involving rats, rabbits, and guinea pigs have demonstrated to us that comparatively large amounts of antimony may kill tiny animals. Rats may undergo lung, heart, liver, and kidney failure before mortality.
- Animals who breathe even in the low level of antimony for an extended period may develop eye irritation, hair loss, and lung damage. Dogs may have heart difficulties even when they are exposed to tiny amounts of antimony.
- Animals that inhaled trace amounts of antimony for several months might suffer reproduction issues.
References
- W. M. Haynes, ed., CRC Handbook of Chemistry and Physics, CRC Press/Taylor and Francis, Boca Raton, FL, 95th Edition, Internet Version 2015, accessed December 2014.
- https://www.britannica.com/science/antimony
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1150–151. ISBN 978-0-08-037941-8.
- https://www.rsc.org/periodic-table/element/51/antimony
- https://www.lenntech.com/periodic/elements/sb.htm
- Tables of Physical & Chemical Constants, Kaye & Laby Online, 16th edition, 1995. Version 1.0 (2005), accessed December 2014.
- https://pubchem.ncbi.nlm.nih.gov/element/Antimony
- He M, Wang N, Long X, Zhang C, Ma C, Zhong Q, Wang A, Wang Y, Pervaiz A, Shan J. Antimony speciation in the environment: Recent advances in understanding the biogeochemical processes and ecological effects. J Environ Sci (China). 2019 Jan;75:14-39. doi: 10.1016/j.jes.2018.05.023. Epub 2018 Jun 14. PMID: 30473279.
- https://chemistrytalk.org/antimony-element/
- Hansell, C. All manner of antimony. Nature Chem 7, 88 (2015). https://doi.org/10.1038/nchem.2134
- Antimony allotropes fabricated on oxide layer of Cu(111): Min Huang,Yu Zhong, Shuangzan Lu, Qinmin Guo, Yinghui Yu
