Amylose Definition
Amylose is a polysaccharide consisting of α-D-glucose units that are linked together by α(1→4) glycosidic bonds.
- An amylose is an essential form of resistant starch that is more resistant to digestion than other starch molecules. The tightly packed helical structure of the molecule aids in its resistance.
- The carbon atoms in glucose are numbered, beginning with carbon 1 at aldehyde that is linked with the 4-carbon of the next molecule, resulting in an α(1→4) glycosidic linkage.
- The number of glucose units in amylose is between 300-3000, but the number can range up to thousands in some cases.
- The amylose chains can exist in three forms; a disordered amorphous conformation or two different helical forms.
- The chain can either bind with itself to form a double helix or with other molecules like iodine and fatty acids.
- The long chains of amylose can more readily crystallize than other forms of glucose, which also increases their resistance to heat and digestive enzymes.
- Amylose occupies about 20-25% concentration in starch, where the glucose molecules are condensed into long chains.
- The increased concentration of amylose in starch decreases the expansion potential and gel strength of the starch.
- Amylose is prevalent in plants as it is a constituent of starch molecules. It was discovered by Meyer and his co-workers in 1940 during their studies on maize.
- It is essential in plant storage as it is less readily digested than amylopectin and requires less space due to its helical structure.
- The chains are broken down by the digestive enzyme α-amylase into maltotriose and maltose.
- Amylose can also be used as a thickener, water binder, and emulsion stabilizer in food and industries.
- Amylose can be detected by the addition of iodine which results in a dark blue-black coloration.
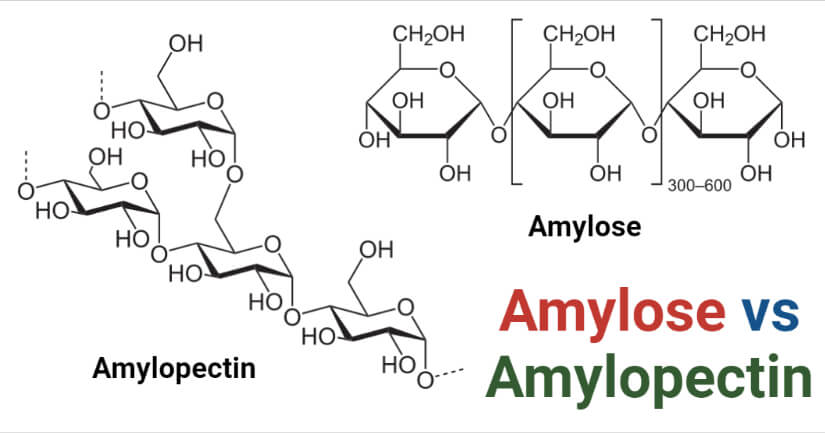
Image Credit: NEUROtiker and NEUROtiker.
Amylopectin Definition
Amylopectin is a type of polysaccharide consisting of a highly branched polymer of α-glucose units.
- The glucose units in amylopectin are linked in a linear chain by α(1→4) glycosidic bonds, and the branching occurs by α(1→6) bonds between 20-30 glucose units.
- The α(1→6) bonds account for about 4-6% of total linkages in the amylopectin molecule. The molecule consists of three different types of chains.
- The A chains are linear chains consisting of a single α(1→6)linkage, whereas the B chains are the chains connected to other chains by α(1→6) linkages. The C chains are the central linear chains consisting of the reducing group (aldehyde group).
- Amylopectin is a component of starch where it accounts for about 80-85% by weight, but the concentration differs depending on the source.
- Even though in most plants, amylopectin accounts for 80% of the starch, 100% amylopectin can be found in the waxy varieties of starches.
- The clustered branches of amylopectin chains are packed together to form the crystalline domains of the starch granules.
- The functional characteristics of the starch-like viscosity, solubility, texture, and gel stability depend on the ratio of amylopectin and amylose.
- The molecular size of amylopectin molecules is much larger than amylose, and they have a clustered branched structure.
- These molecules usually form soft pastes of gels instead of hard clumps that are characterized by clarity, stability, and resistance to aging.
- Even though amylopectin is the main component of starch, there is no effective method for the direct estimation of amylopectin, and most of the studies on amylopectin are dependent on enzymatic and instrumental techniques.
- These methods have indicated that starches of different botanical sources have different branch chain lengths and distribution of amylopectin.
Read Also: Alkali vs Base- Definition, 7 Key Differences, Examples
12 Key differences (Amylose vs Amylopectin)
| Characteristics | Amylose | Amylopectin |
| Definition | Amylose is a polysaccharide consisting of α-D-glucose units that are linked together by α(1→4) glycosidic bonds. | Amylopectin is a type of polysaccharide consisting of a highly branched polymer of α-glucose units. |
| Molecular size | The molecular size of amylose is less than that of amylopectin. | Amylopectin molecules are larger than amylose molecules. |
| Concentration in waxy starch | Minimal to no amylose is found in waxy starches. | Waxy starches can contain up to 100% amylopectin. |
| Linkages | The glucose units in amylose are linked together by α(1→4) glycosidic bonds. | The glucose units in amylopectin are linked together by α(1→4) glycosidic bonds and α(1→6) linkages. |
| Branching | The glucose units in amylose are arranged in a linear chain with no branching. | The glucose units are amylopectin are arranged in both linear and branched forms. |
| Starch | Amylose content in starch ranges between 20-25%. | Amylopectin content in starch ranges between 80-85%. |
| Solubility in water | Amylose is less soluble in water. | Amylopectin is more soluble in water. |
| Gel formation | Amylose doesn’t form a gel after the addition of water. | Amylopectin forms a gel after the addition of water. |
| Addition of iodine | Amylose results in a blue/black coloration after the addition of iodine. | Amylopectin gives a reddish-brown coloration after the addition of iodine. |
| Resistance to digestive enzymes | Amylose is comparatively more resistant to digestive enzymes due to its helical structure. | Amylopectin is comparatively less resistant to digestive enzymes due to the branched structure. |
| Crystallization | The linear chains of amylose crystallize more readily than amylopectin. | The short branched chains of amylopectin cannot crystalline as readily. |
| Function | Amylose in starch enables the storage of starch in plants. | Amylopectin in starch allows the utilization of starch as a source of energy. |
References and Sources
- A. Gunaratne, H. Corke. Starch: Analysis of Quality. Encyclopedia of Food Grains (Second Edition). 2016. Pages 198-207. https://doi.org/10.1016/B978-0-12-394437-5.00092-9.
- C.V. Ratnavathi, V.V. Komala. Chapter 1 – Sorghum Grain Quality. Sorghum Biochemistry, 2016. Pages 1-61. https://doi.org/10.1016/B978-0-12-803157-5.00001-0.
- 3% – https://www.difference.wiki/amylose-vs-amylopectin/
- 2% – https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/amylopectin
- 2% – https://www.sciencedirect.com/science/article/pii/S0268005X12002974
- 1% – https://www.sciencedirect.com/topics/engineering/iodine-number
- 1% – https://www.sciencedirect.com/topics/engineering/amylopectin
- 1% – https://www.sciencedirect.com/science/article/pii/S0144861719301857
- 1% – https://www.researchgate.net/publication/275662036_Determination_of_amyloseamylopectin_ratio_of_starches
- 1% – https://www.academia.edu/12805077/Influence_of_Phase_Behavior_and_Miscibility_on_Mechanical_Thermal_and_Micro_Structure_of_Soluble_Starch_Gelatin_Thermoplastic_Biodegradable_Blend_Films
- 1% – https://treehozz.com/what-polysaccharides-are-not-digestible-by-humans
- 1% – https://en.wikipedia.org/wiki/Amylose
- <1% – https://www.healthline.com/health/food-nutrition/is-maltodextrin-bad-for-me
- <1% – https://vivadifferences.com/difference-between-amylose-and-amylopectin/
- <1% – https://en.wikipedia.org/wiki/Polymorphism_%28biology%29
- <1% – https://chem.libretexts.org/Courses/Sacramento_City_College/SCC%3A_Chem_309_-_General_Organic_and_Biochemistry_(Bennett)/Text/14%3A_Carbohydrates/14.7%3A_Polysaccharides

Thank for your help